Targeting disease origin over time: suppressing the source, not the effector
Note: The video and audio linked above were generated with the assistance of AI. Clinical accuracy has been reviewed, but no AI-generated content can be guaranteed to be fully error-free.
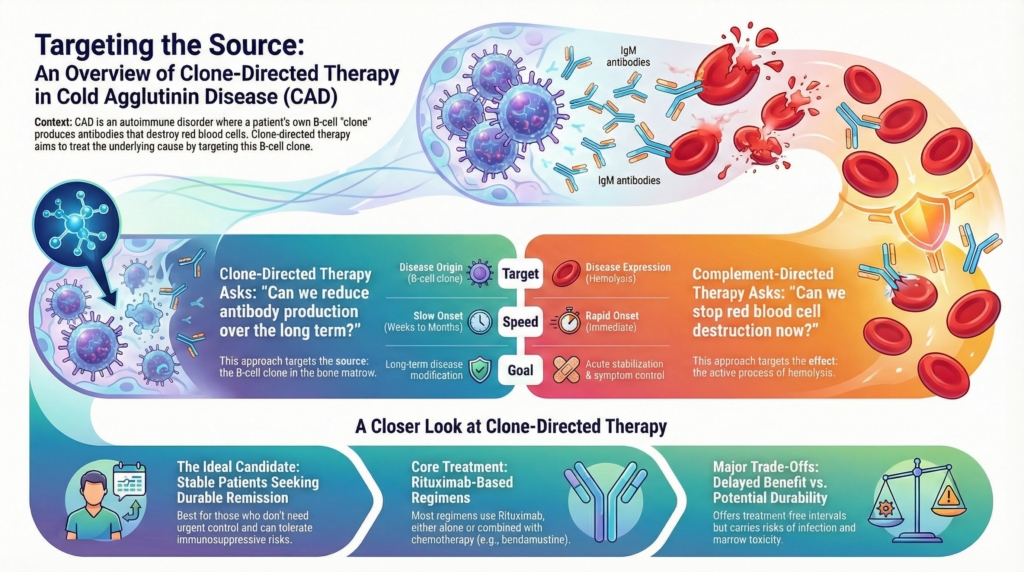
Why this spoke matters
Cold agglutinin disease is now understood as a clonal disorder with immune consequences. Primary CAD represents a bone-marrow–based, indolent B-cell lymphoproliferative disease that secondarily causes hemolysis through production of a monoclonal IgM autoantibody that activates complement.1
Complement-directed therapy and clone-directed therapy intervene at different points along this disease pathway. Complement inhibition acts at the effector phase to suppress ongoing hemolysis, whereas clone-directed therapy targets the upstream source of pathogenic antibody production. Because these approaches address different mechanisms, they serve different clinical purposes and should not be conflated, as emphasized in contemporary reviews of CAD management.2
This section focuses on clone-directed therapy: what it aims to accomplish, when it may be appropriate, and why its benefits and limitations differ in important ways from complement inhibition.3
The biological target
In primary CAD, the pathogenic IgM antibody is produced by an underlying indolent B-cell clone, most often confined to the bone marrow and frequently not meeting diagnostic criteria for overt lymphoma.4
Well-described features of this clonal substrate include:5
- monoclonal IgM production, typically κ-restricted
- an indolent B-cell lymphoproliferative disorder in virtually all well-studied primary CAD marrows
- clonal persistence over years
- a variable, often weak relationship between clone size and the clinical severity of hemolysis
Clone-directed therapy is therefore aimed at this B-cell population rather than at the downstream hemolytic process itself.6
What clone-directed therapy attempts to accomplish
Clone-directed therapy seeks to reduce or eliminate antibody production by suppressing or eradicating the responsible B-cell clone.
Its intended effects include:7
- reduction in pathogenic IgM production
- downstream reduction in complement activation
- improvement in hemolysis over time
- the potential for prolonged treatment-free intervals
When effective, this approach can lead to meaningful hematologic improvement and, in some patients, prolonged treatment-free intervals, particularly with combination rituximab-based regimens.8
Unlike complement inhibition, clone-directed therapy is explicitly aimed at disease origin rather than disease expression, with the goal of modifying the long-term course rather than providing immediate control.9
This approach aligns with contemporary reviews that describe primary CAD as a clonal B-cell disorder in which therapy is directed at reducing pathogenic IgM production, rather than directly inhibiting complement activation.10
What clone-directed therapy does not do
Clone-directed therapy does not provide immediate disease control.
It does not:11
- rapidly suppress ongoing hemolysis
- reliably stabilize patients during acute exacerbations
- predictably improve anemia within days to weeks
- function as an emergency intervention
For this reason, rituximab-based regimens are generally not relied upon when the dominant clinical need is urgent stabilization of active hemolysis or acute disease exacerbation.12
Clinical benefit, when it occurs, typically evolves over weeks to months, reflecting the time required to suppress the pathogenic B-cell clone and reduce IgM production, with subsequent downstream effects on complement activation and hemolysis.13
Clone-directed therapies
Common clone-directed regimens in CAD
In clinical practice, clone-directed therapy in primary CAD relies predominantly on rituximab-based regimens, adapted from treatment strategies used for indolent B-cell lymphoproliferative disorders rather than from aggressive lymphoma or plasma-cell paradigms.14
The most commonly used approaches include:
Rituximab monotherapy15
- anti-CD20 monoclonal antibody
- overall response rates in the range of approximately 45–60%
- relapse is reported in 57%-89% of patients who respond
- median response durations typically in the range of approximately 6–11 months
- often considered in older patients or those unable to tolerate combination therapy
Rituximab plus fludarabine16
- purine-analog–based chemoimmunotherapy
- higher response rates and deeper remissions than rituximab alone
- substantial immunosuppression and infection risk
- now used selectively rather than routinely, largely due to immunosuppression and infection risk
Rituximab plus bendamustine17
- alkylator-based chemoimmunotherapy
- favored in many centers for patients fit for cytotoxic therapy
- high response rates with multi-year remissions reported in prospective cohorts
- improved tolerability compared with fludarabine-based combinations, though still immunosuppressive
These regimens are selected not to eradicate bulk tumor mass, but to sufficiently suppress the IgM-producing clone so as to reduce pathogenic antibody production over time.18
In many centers, rituximab–bendamustine has become the preferred combination regimen when cytotoxic therapy is appropriate, because it offers high response rates with a more favorable tolerability profile in typical CAD patients.
Therapies generally not used in primary CAD
It is equally important to understand which therapies are generally ineffective or inappropriate in primary cold agglutinin disease, because several interventions commonly used in other autoimmune hemolytic anemias do not translate well to cold antibody–mediated disease.19
Glucocorticoids have limited efficacy in CAD and are generally discouraged as routine therapy. Responses are uncommon, often incomplete, and may require high doses that expose patients to disproportionate toxicity.20
Aggressive lymphoma regimens (for example, R-CHOP–like therapy) are not part of routine primary CAD management and should be reserved for patients who meet independent criteria for treatment of an overt lymphoma.21
Therapies designed for plasma cell neoplasms or high-grade malignancies are not standard approaches in primary CAD, where the underlying clone is typically indolent and marrow-based.22
This distinction reinforces that clone-directed therapy in CAD is targeted suppression of an indolent marrow clone, not escalation along a hematologic malignancy ladder.23
A practical corollary is that apparent “AIHA” that fails to respond to glucocorticoids should prompt reconsideration of mechanism, including evaluation for cold antibody–mediated disease.
Choosing among clone-directed options
Selection among rituximab-based regimens is individualized and reflects a balance between the desired depth and durability of response and the patient’s tolerance for immunosuppression and cytotoxic toxicity. Selection among rituximab-based regimens depends on:24
- patient age and comorbidity burden
- tolerance for immunosuppression
- desire for finite therapy versus risk minimization
- need for depth and durability of response
- coexistence of lymphoma-related indications
There is no single best regimen for all patients. Decisions typically involve trading greater likelihood of prolonged remission with combination therapy against higher risks of infection, cytopenias, and marrow toxicity, particularly in older or medically frail individuals.25
Temporal profile of response
The tempo of response is determined by biology, not drug potency, so it is delayed and variable. In studies of rituximab monotherapy, clinical responses typically emerge over several weeks to a few months, with remissions that are often limited in duration compared with combination regimens. This reflects the time required to suppress pathogenic IgM production, rather than to immediately interrupt complement-mediated hemolysis.26
In clinical series summarized in recent ASH and expert reviews, the median time to response with rituximab is approximately 1.5–3 months, with remission durations typically measured in months and relapse common within a year after B-cell repopulation.27
Combination regimens, such as rituximab with fludarabine or bendamustine, can produce deeper and more durable responses, but they follow the same fundamentally gradual trajectory. Improvement evolves as antibody production declines, rather than as an immediate pharmacologic effect on hemolysis.28
Typical response characteristics include:29
- gradual decline in IgM levels
- delayed improvement in hemolysis markers
- hemoglobin recovery over months
- heterogeneity in depth and durability of response
In practice, response is tracked with hemoglobin and hemolysis markers, while IgM level (when available) can provide supportive context for trajectory. Lack of improvement at a few weeks is not failure. The key question is whether there is a directional shift by 8–12 weeks, and whether the patient remains clinically stable while the regimen has time to work.
Rituximab depletes circulating and tissue CD20-positive B cells, but it does not eliminate all upstream precursors or fully eradicate a marrow-based clone. Over time, B-cell repopulation can restore IgM production and hemolysis may recur. Combination regimens can produce deeper and longer remissions because they suppress a broader portion of the clonal compartment, at the cost of greater cytopenias and infection risk. This is why durability differs across regimens even when the mechanistic goal is the same: reducing pathogenic IgM production.

Durability and disease modification
Clone-directed therapy offers the possibility—but not the guarantee—of durable clinical benefit. By suppressing the pathogenic B-cell clone, these approaches aim to reduce antibody production over time rather than to provide immediate disease control.
Potential advantages demonstrated in clinical series include:30
- prolonged remission without continuous therapy
- reduced dependence on ongoing pharmacologic suppression during remission
- alignment with disease-modifying goals
Reported durability varies substantially by regimen. In published series, median response durations are approximately 6–11 months with rituximab monotherapy, whereas combination regimens have been associated with multi-year remissions, extending beyond five years in a proportion of patients.31
However, responses are heterogeneous, and relapse remains common, particularly after rituximab monotherapy. Suppression of the B-cell clone does not reliably translate into permanent disease eradication, and long-term follow-up confirms that CAD remains a chronic condition in most patients.32
Who may benefit most
Clone-directed therapy is most appropriate for patients with:33
- relatively stable disease where immediate control is not required
- preference for finite treatment rather than chronic therapy
- acceptable tolerance for immunosuppression
- clonal features suggesting treatment responsiveness, when present (although reliable predictive biomarkers are limited)
- coexisting indications to treat the underlying lymphoproliferative disorder
It is particularly considered when long-term disease modification is a central priority and the clinical situation allows time for a slower-acting regimen to work.34
Risks and trade-offs
Clone-directed therapy carries risks that differ fundamentally from those of complement inhibition.
Key considerations documented in trials include:35
- immunosuppression and infection risk
- cytopenias and marrow toxicity
- delayed onset of benefit
- variable response rates
- risk of overtreatment in otherwise indolent disease
These trade-offs must be weighed carefully against disease burden, comorbidities, and patient priorities, particularly in older patients or those with limited physiologic reserve.36
Clone-directed therapy vs complement-directed therapy
Clone-directed therapy and complement-directed therapy target different mechanisms and address different clinical needs.
Clone-directed therapy asks:
“Can we reduce pathogenic antibody production over time?”
Complement-directed therapy asks:
“Can we suppress hemolysis now?”
This distinction is made explicitly in contemporary CAD management reviews.37
The approaches are not interchangeable. Complement inhibitors do not modify the underlying clone, and rituximab-based regimens are not appropriate for acute stabilization of active hemolysis.38
Recent ASH guidance explicitly frames rituximab-based therapy as targeting pathogenic antibody production with delayed clinical benefit, whereas complement inhibition provides rapid control of hemolysis without modifying the underlying clone.39
Combination and sequencing
In selected patients, clone-directed and complement-directed therapies may be combined or sequenced, depending on clinical priorities. This approach mirrors contemporary expert recommendations, which describe complement inhibition as a potential bridge to slower, clone-directed strategies in selected patients.40
Conceptual strategies described in the literature include:41
- complement inhibition for rapid control of hemolysis followed by rituximab-based therapy for longer-term modulation
- clone-directed therapy alone in stable disease with low immediate risk
- complement inhibition alone when disease control is needed but clone suppression is undesirable
The optimal sequence is individualized and goal-dependent, and no randomized trials currently define a single preferred strategy.
When clone-directed therapy fails or responses are not durable, clinicians often reconsider complement inhibition, alternative rituximab-based regimens, or observation, depending on disease tempo and patient priorities.
Explicit limits of this strategy
Clone-directed therapy is not ideal when:42
- rapid disease control is required
- disease burden is clearly driven by complement activity rather than clone size
- risks of immunosuppression outweigh potential benefit
- disease is sufficiently mild that observation is appropriate
Understanding these limits helps prevent misapplication of a slow but potentially durable approach.
Explicit principle
Clone-directed therapy targets disease origin, not disease expression.
It offers the possibility of longer-term remission but not rapid control. Mastery of this strategy lies in matching its slower tempo and immunologic risks to patients whose disease course, priorities, and stability make disease modification a realistic and appropriate goal.
Reflect and Apply
A 74-year-old with primary CAD has Hb 7.9 g/dL and is transfusion-dependent. You start complement inhibition and within 2 weeks Hb rises to 10.8 g/dL with symptom improvement. She asks, “Does this mean my disease is cured, and can I stop soon?”
- What do you say about control vs modification?
- Under what conditions would you discuss clone-directed therapy?
- What trade-offs would you name (tempo, immunosuppression, durability, goals)?
What trade-offs would you name (tempo, immunosuppression, durability, goals)?
Evidence anchor: clone-directed regimens studied in primary CAD
Summary derived from key prospective trials, long-term follow-up studies, and systematic reviews
Rituximab monotherapy
- Regimen: rituximab 375 mg/m² IV weekly × 4
- Overall response: ~50–60%
- Median response duration: ~6–11 months
- Relapse common; retreatment often effective
Evidence base:
- Berentsen S et al., prospective monotherapy cohort, ORR ~50%.43
- Meta-analysis of rituximab in AIHA (including cold types) reporting ORR ~57%44
Rituximab + fludarabine
- Higher response rates and longer remissions than rituximab alone
- Substantial immunosuppression and infection risk
Evidence base:
- Berentsen S et al., prospective multicenter trial, ORR 76%, long response duration, notable toxicity (grade 3-4 hematologic toxicity occurred in 41%).45
Rituximab + bendamustine
- Deep and durable responses in severe or refractory CAD
- Finite 4‑cycle course with higher upfront myelosuppression than rituximab alone, but acceptable safety in fit patients.
Evidence base:
- Berentsen S et al., prospective, nonrandomized multicenter trial, rituximab plus bendamustine reported to induce response in 70% of adults with cold agglutinin disease, with 91% of responders maintaining response at median 32 months.46
Interpretive note
These data are derived largely from non-randomized trials and observational cohorts; no head-to-head randomized comparisons exist.
Guideline perspective: clone-directed therapy in CAD
Based on international guideline and consensus statements
Guideline organizations referenced
- British Society for Haematology (BSH) guideline on diagnosis and management of primary autoimmune hemolytic anemia47
- Expert consensus reviews in Hematology and British Journal of Haematology
- International consensus and review articles on CAD management
Shared guidance themes
- Rituximab-based therapy recommended when treatment is indicated
- Indications include:
- symptomatic anemia
- severe cold-induced circulatory symptoms
- transfusion dependence
- Combination chemo-immunotherapy reserved for:
- severe disease
- relapsed disease
- patients fit for cytotoxic therapy
What guidelines emphasize
- Individualized decision-making
- Focus on disease modification and durable remission, not rapid hemolysis control (which is better addressed by complement inhibition).
- Avoidance of treatment in mild or asymptomatic disease
What guidelines do not require
- Eradication of the B-cell clone
- Treatment of fatigue alone
- A single preferred regimen for all patients
Test your thinking
A short, judgment-focused quiz on clone-directed therapy in cold agglutinin disease.
