Labs
The following is the complete blood count (CBC) at the time of admission:
| WBC (109/L) | Hb (g/dL) | MCV (fL) | PLT (109/L) |
|---|---|---|---|
| 9.7 | 7.4 | 92 | 259 |
What’s what: WBC, white blood cell count; Hb, hemoglobin; MCV, mean cell volume; MCHC, mean cellular hemoglobin concentration; RDW-SD, red cell distribution width-standard deviation; platelets, PLT; Normal values: WBC 5-10 x 109/L, RBC 4-6 x 1012/L, Hb 12-16 g/dL, Hct 35-47%, MCV 80-100 fL, MCHC 32-36 g/dL, RDW-SD < 45 fL, platelets (PLT) 150-450 x 109/L
PT and aPTT results:
| PT (seconds) | aPTT (seconds) |
|---|---|
| 13.8 | 81.6 |
PT is minimally elevated. For all intents and purposes, we can approach this case as an isolated elevation in the aPTT.
Let’s look at how a PT and aPTT are carried out:
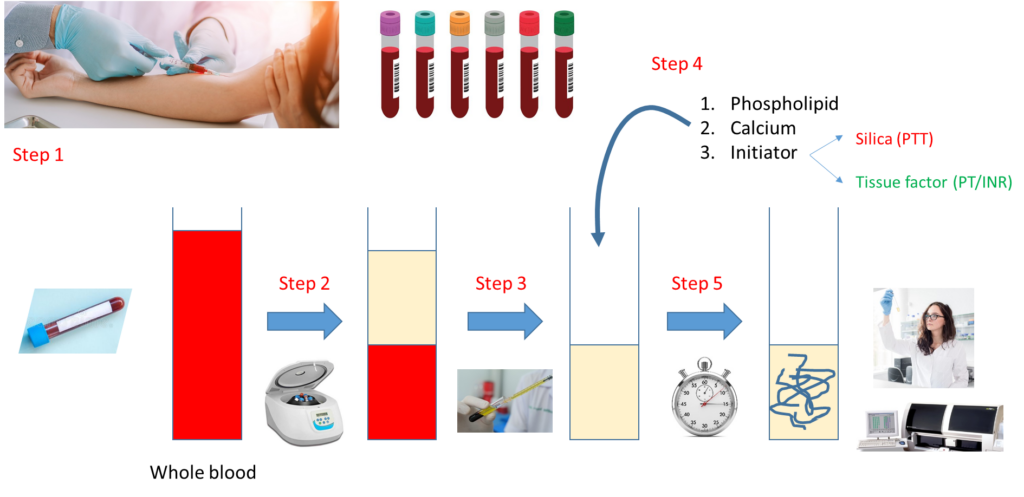
Notes:
- Step 1 – Draw whole blood from a blood vessel (typically an arm vein) into a blue top tube containing the anticoagulant, sodium citrate.
- Step 2 – Spin the liquid blood sample in a centrifuge so that red cells layer on the bottom, plasma on the top.
- Step 3 – Pipette plasma (top layer) into a clean, empty test tube.
- Step 4 – Add phospholipid, calcium and activator (tissue factor for PT, silica for aPTT).
- Step 5 – Incubate at 37 degrees C and measure time to clot formation, either with machine or more rarely by vision.
Note that the major difference between the PT and aPTT is the nature of the activator added to the sample.
What are possible causes of an isolated elevation in the aPTT (i.e., the PT is normal)?
Back to the patient. He presents with bilateral arm swelling and a high aPTT. What are the two most likely causes of the arm swelling?
A Doppler ultrasound of the upper extremities was ordered in the emergency room.
These are the results:
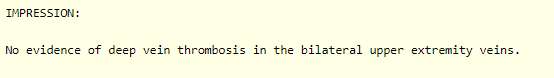
Therefore, the swelling in the arms is most certainly secondary to hematoma.
Further work-up of this case requires some knowledge of the clotting cascade. Let’s review the basics:
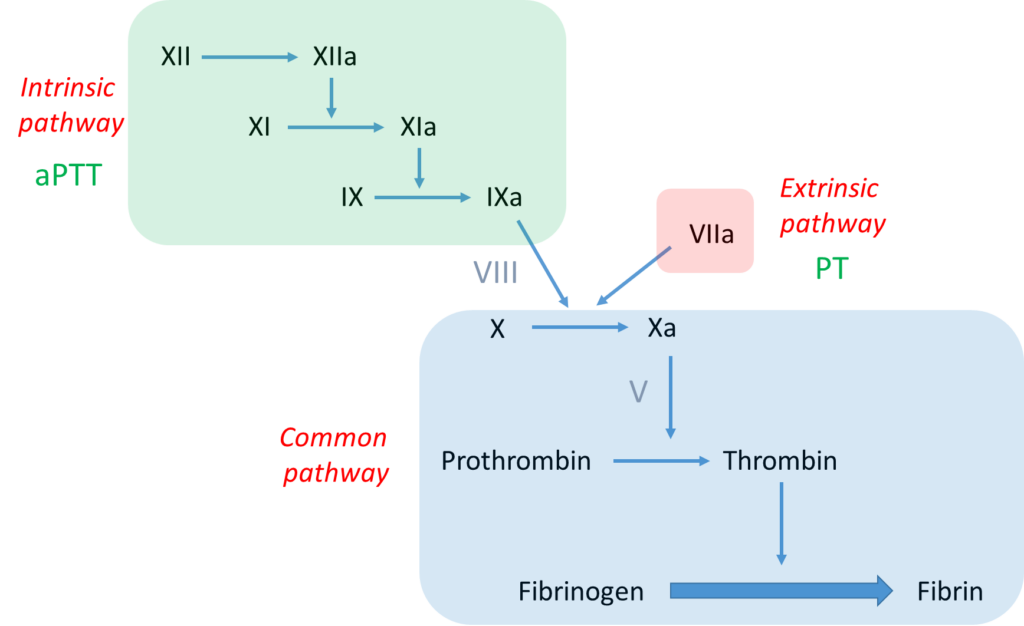
Notes:
- The bottom line is formation of an insoluble fibrin plug, which, along with platelets, stems blood loss.
- Fibrin is derived from thrombin-mediated cleavage of soluble fibrinogen (which is a structural protein).
- Thrombin (which is a type of enzyme called a serine protease) is formed when another serine protease, activated factor X (FXa), cleaves prothrombin.
- Two pathways may activate factor X:
- The intrinsic pathway, which consists of a series of linked reactions involving serine proteases FXII, FXI and FIX, and cofactor FVIII.
- The extrinsic pathway, which consists of tissue factor-mediated activation of FVII (FVIIa).
- In vivo, the clotting cascade is always initiated by tissue factor activation of FVII (extrinsic pathway) and amplified by the intrinsic pathway via cross-talk (FVIIa activates FIX) and feedback (thrombin activates FXI and FVIII) mechanisms.
- aPTT measures the integrity of the intrinsic pathway.
- PT/INR measures the integrity of the extrinsic pathway.

According to the scheme above, an isolated elevation in the aPTT indicates a deficiency of or inhibitor against a clotting factor in the intrinsic pathway, namely FXII, FXI, FIX or FVIII.
Heparin is often considered to be a cause of isolated aPTT elevation. Heparin inhibits components of the intrinsic, extrinsic and common pathways, so in theory it should raise both the PT and aPTT. In fact, heparin does increase the PT, but the degree of change is small (5-10%) and at therapeutic doses barely noticeable:
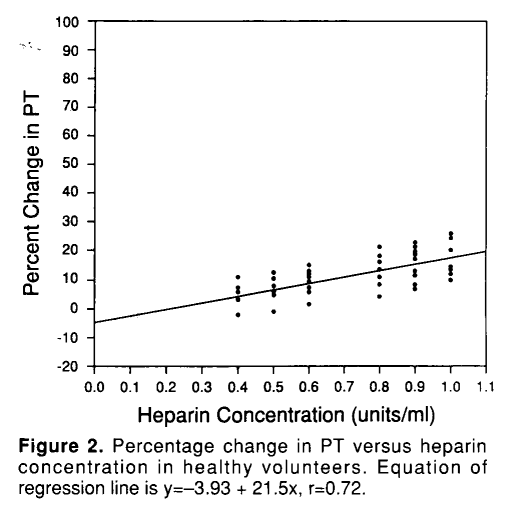
This patient provides no history of heparin exposure. But there are cases, especially in hospitalized patients, where accidental or unidentified heparin exposure may be an issue.
Let’s consider each assay in turn:
Thrombin time
- Measures the time required for formation of a stable clot, following addition of thrombin to citrated plasma.
- Provides the measure of conversion of fibrinogen to fibrin and therefore an estimation of the quantity of fibrinogen.
- Causes of prolonged thrombin time include:
- Afibrinogenemia
- Hypofibrinogenemia
- Dysfibrinogenemia
- Factors that inhibit thrombin-mediated conversion of fibrinogen to fibrin:
- Heparin
- Hirudin
- Other direct thrombin inhibitors
- Autoantibodies, for example in multiple myeloma
- Fibrin degradation products, for example in disseminated intravascular coagulation (these inhibit thrombin-mediated cleavage of fibrinogen)
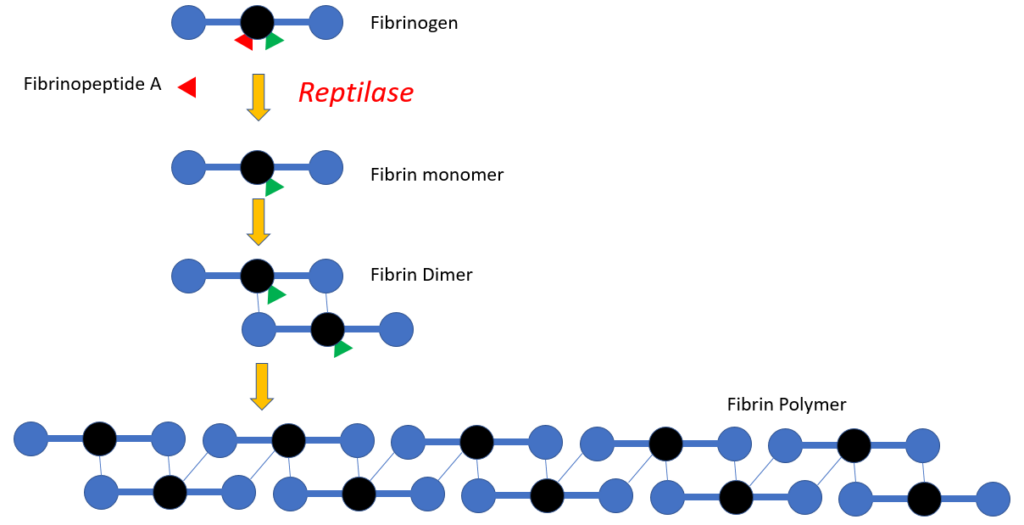
Reptilase time
- A venom-based functional plasma clotting assay based on the enzymatic activity of batroxobin, the thrombin-like proteolytic enzyme isolated from the venom of the South American pit viper.
- Unlike thrombin, which cleaves fibrinopeptides A and B from fibrinogen, batroxobin splits specifically fibrinopeptide A, converting fibrinogen into fibrin monomers. The resulting fibrin monomers polymerize spontaneously and lead to the formation of a stable fibrin clot.
- The time, starting from the addition of batroxobin to the plasma sample, until clot formation is the reptilase time and is given in seconds.
- The reptilase time is independent of thrombin generation disturbances or disturbances in the action of thrombin on fibrinogen. Therefore, it can be used to confirm heparin contamination or to obtain similar information as with the thrombin clotting time in heparinized and hemophiliac patients.
- The clotting time obtained by RT is independent of thrombin generation disturbances and disturbances of thrombin–fibrinogen interaction and is not affected by anticoagulants such as heparin and hirudin.
- Causes of prolonged reptilase time include:
- Afibrinogenemia
- Hypofibrinogenemia
- Dysfibrinogenemia
- Factors that inhibit conversion of fibrinogen to fibrin:
- Especially fibrin degradation products
- Not sensitive to antithrombin inhibitors
Which of the following is true (more than one answer may apply)?
Back to the patient. What would you like to test for next?
A mixing study is a reasonable next step in a patient with unexplained elevation in PT and/or aPTT. What is a mixing study?
Let’s review the basics of the mixing study:
Notes:
- Mixing study involves mixing patient plasma and pooled normal plasma in 1:1 ratio.
- The resulting mixture is then processed for clotting time (PT or aPTT) immediately (time 0) and then after 2 hours of incubation.
- A clotting factor must drop below about 30% before the PT or PTT begins to prolong.
- Therefore, even if a clotting factor is completely missing (i.e. 0% level), the 1:1 mix will restore it to 50% (equal volume of plasma containing 0% of the factor and normal pooled plasma containing 100% of the factor), more than enough to restore a normal PT or aPTT (the example above shows 10% activity of imaginary factor Y in patient plasma, with 1:1 mix resulting in 60% activity).
- However, if there is an inhibitor or antibody to a clotting factor, the inhibitor will carry over from patient to normal plasma in the mix and interfere with factor activity, leading to continued prolongation of the PT or aPTT.
Mixing study (50:50 mix) results
(baseline aPTT 76.7, PT 13.1)

What is the result of the above mixing study?
Mixing study (50:50 mix) results


What type of inhibitor causes equal prolongation of aPTT at time 0 and after 2 h incubation?
Click for AnswerIf the PT, and not the aPTT was prolonged and the prolongation failed to correct with the 1:1 mix, what factor would you predict is being inhibited?
To summarize at this point, we have an elevated aPTT (and normal PT) in a patient with recent rapid onset of bilateral arm swelling with ecchymoses, and a positive inhibitor screen (mixing study), indicating the presence of an inhibitor against one or more factors (almost certainly a single factor) in the intrinsic pathway.
Which factor level would you prioritize investigating?
the patient’s factor VIII level was <1% (normal 50-150%)
What is the diagnosis?
Are you surprised there is no family history of bleeding?
About 50% of cases of acquired hemophilia A are secondary to an underlying medical condition. Which of the following conditions may be associated with acquired hemophilia A?
Acquired and congenital hemophilia A have a similar bleeding phenotype, with one notable exception.

Compared to congenital hemophilia A, bleeding at this site is infrequent in acquired hemophilia A
Click for AnswerLet’s consider the anemia for a moment. The patient’s Hb is 7.3

