When the A1c Doesn’t Fit the Patient
When the A1c Doesn’t Fit the Patient
An unexpected clue to compensated hemolysis
By William C. Aird, MD
Case Presentation
A 74-year-old woman is referred to hematology for evaluation of possible hemolysis.
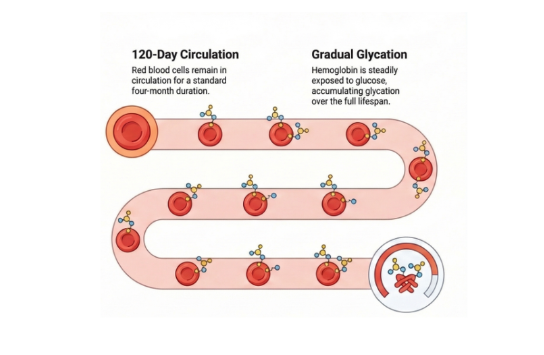
Her endocrinologist noted that her hemoglobin A1c appeared lower than expected given her glucose readings. This discrepancy raised concern that red cell turnover might be increased.
Hemoglobin A1c reflects glycation of hemoglobin that accumulates over the lifespan of circulating red blood cells. If red cells are destroyed prematurely, they spend less time in circulation and accumulate less glycation, causing the A1c to underestimate true glycemic exposure.
She reports feeling well. She denies fatigue, dyspnea, dark urine, jaundice, or symptoms triggered by cold exposure such as acrocyanosis or Raynaud phenomenon.
Past medical history includes:
- type 1 diabetes mellitus
- hypercholesterolemia
- lymphocytic colitis
- diabetic neuropathy
- diabetic retinopathy
- gastroesophageal reflux disease
There is no family history of hematologic disease.
Physical examination is unremarkable.
Initial Laboratory Findings
Laboratory studies demonstrate evidence of hemolysis:
| Test | Result |
|---|---|
| Hemoglobin | 12.1 g/dL |
| Reticulocyte count | Elevated |
| Haptoglobin | <10 mg/dL |
| LDH | Elevated |
| Direct antiglobulin test | C3d positive, IgG negative |
| Cold agglutinin screen | Positive |
Example blood bank report:

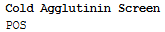
Peripheral smear shows red cell clumping, and the MCHC is elevated.
Example peripheral smear:
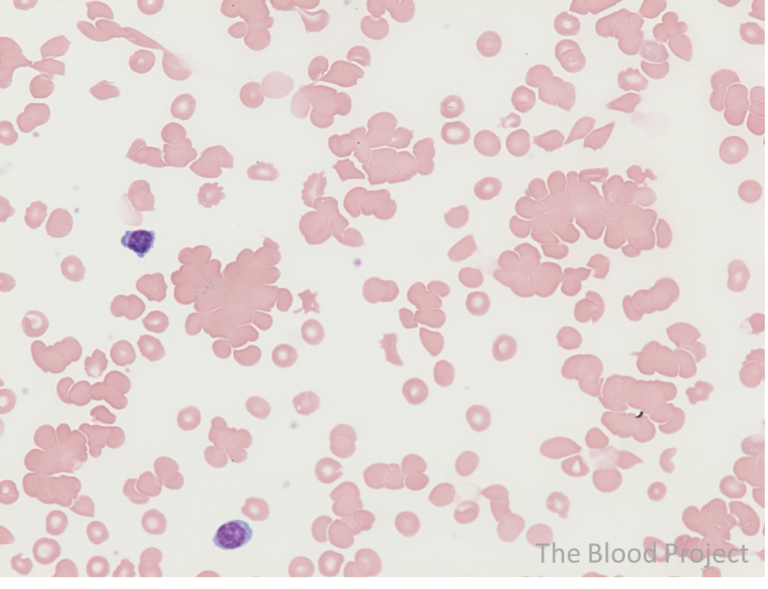
Cold agglutinins cause red cells to aggregate at lower temperatures. Automated analyzers may interpret clumped cells as single larger cells, which can artifactually elevate the MCHC. In this context, laboratory artifact itself becomes a diagnostic signal.
What process best explains this laboratory pattern?
Explanation
This laboratory pattern is characteristic of hemolysis.
Key features include:
- elevated reticulocyte count, reflecting increased marrow response to red cell destruction
- low haptoglobin, due to binding of free hemoglobin released during hemolysis
- elevated LDH, released from lysed red cells
Together, these findings indicate increased red cell destruction with compensatory erythropoiesis.
Which of the following findings most strongly supports the diagnosis of cold agglutinin disease?
Diagnostic Evaluation
Because cold agglutinin disease can be associated with an underlying clonal B-cell lymphoproliferative disorder, the patient underwent additional evaluation.
Bone marrow biopsy demonstrated:
- hypercellular marrow
- mild erythroid-predominant trilineage hematopoiesis
- no evidence of lymphoma or plasma cell neoplasm
The purpose of the marrow examination was to exclude an underlying lymphoproliferative disorder, which would change both the diagnosis and management strategy.
In this case, no secondary cause was identified.
These findings are most consistent with primary cold agglutinin disease.
Clinical Course
Despite laboratory evidence of hemolysis, the patient remained asymptomatic with a normal hemoglobin level.
Laboratory follow-up demonstrated:
| Test | Result |
|---|---|
| Hemoglobin | 12.1 g/dL |
| Reticulocyte count | Elevated |
| Haptoglobin | <10 mg/dL |
Which of the following best describes this patient’s current hematologic state?
Explanation
In compensated hemolysis:
- red cell destruction is increased
- the bone marrow increases erythropoiesis sufficiently to maintain hemoglobin
Typical findings include:
- elevated reticulocyte count
- low haptoglobin
- normal hemoglobin
Patients may remain stable for years without requiring therapy.
Which of the following is the most appropriate management strategy for this patient?
Explanation
Treatment of cold agglutinin disease is guided by disease burden, not by laboratory findings alone.
Therapy is typically reserved for patients with:
- symptomatic anemia
- transfusion dependence
- severe hemolysis
- cold-induced circulatory symptoms (e.g., acrocyanosis)
This patient has compensated hemolysis without symptoms, placing her below the treatment threshold.
Management therefore consists of:
- observation
- periodic laboratory monitoring
- counseling about cold exposure
Teaching Points
- Unexpected laboratory discrepancies can reveal hidden physiology.
A mismatch between glucose readings and hemoglobin A1c may signal shortened red cell survival. - The DAT pattern provides mechanistic information.
C3d positivity with negative IgG strongly suggests complement-mediated hemolysis such as cold agglutinin disease. - Laboratory artifacts can be diagnostic clues.
Red cell clumping and elevated MCHC on automated analyzers may reflect cold agglutinin–mediated agglutination. - Treatment decisions in CAD are driven by clinical burden.
Patients with compensated hemolysis and no symptoms are typically managed with observation.
Why this case matters
Cold agglutinin disease is often imagined as a dramatic hemolytic disorder with severe anemia and cold-induced symptoms. In reality, some patients live for years with quiet, compensated hemolysis.
In this patient, the diagnosis emerged from a subtle clue:
A hemoglobin A1c that did not match the patient’s glucose profile.
Recognizing such discrepancies can reveal underlying hematologic processes and prevent both missed diagnoses and unnecessary treatment.
