When oxygen becomes scarce, should biology prioritize loading in the lung or unloading in tissues?
Note: The video and audio linked above were generated with the assistance of AI. Clinical accuracy has been reviewed, but no AI-generated content can be guaranteed to be fully error-free.
Introduction
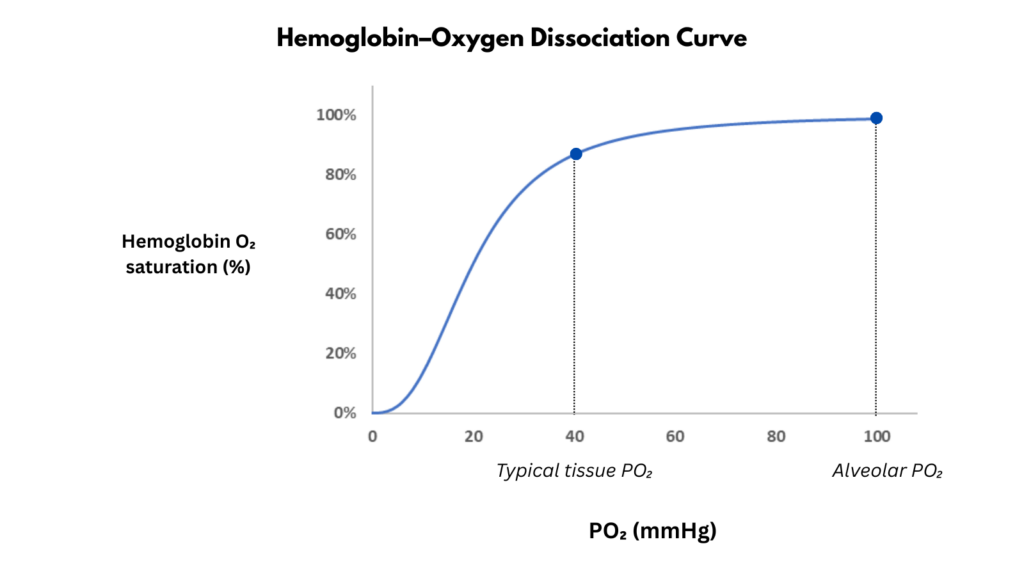
The oxygen dissociation curve is one of the most recognizable graphs in physiology. It describes how hemoglobin saturation varies as the partial pressure of oxygen (PO₂) changes. At high PO₂ in the lung, hemoglobin approaches full saturation. At lower PO₂ in peripheral tissues, oxygen dissociates from hemoglobin and becomes available for cellular metabolism.
The curve reflects an essential property of hemoglobin: cooperative oxygen binding. Because of this cooperativity, the relationship between PO₂ and hemoglobin saturation is sigmoidal rather than linear. In the steep middle portion of the curve, relatively small changes in PO₂ produce large changes in oxygen saturation, facilitating oxygen release where it is needed.
Yet the oxygen dissociation curve describes only one component of a much larger physiological system of oxygen transport.
At the level of the organism, oxygen transport depends on both the delivery of oxygen to tissues and the extraction of oxygen by those tissues. Oxygen delivery reflects the amount of oxygen carried in arterial blood and the rate at which blood flows through the circulation. Oxygen consumption reflects the amount of oxygen removed from blood as it passes through the microcirculation.
Oxygen transport at the system level
Oxygen deliveryDO2=Q×Ca
where
• DO2 = oxygen delivery
• Q = cardiac output
• Ca = arterial oxygen content
Oxygen consumption (Fick principle)VO2=Q×(Ca−Cv)
where
• VO2 = oxygen consumption
• Cv = venous oxygen content
Arterial oxygen contentCa≈1.34×Hb×Sa
where
• Hb = hemoglobin concentration
• Sa = arterial oxygen saturation
Key implication
Hemoglobin affinity affects oxygen transport by influencing:
- arterial oxygen loading in the lung
- oxygen unloading in tissues
PO₂ (the partial pressure of dissolved oxygen in plasma) is the independent variable that governs oxygen binding to hemoglobin. Hemoglobin saturation is the dependent variable, reflecting the proportion of hemoglobin molecules bound to oxygen at a given PO₂.
Hemoglobin affinity refers to how readily hemoglobin binds oxygen and is defined by the degree of hemoglobin saturation at a given PO₂.1
Hemoglobin affinity affects oxygen transport in two ways. By influencing arterial saturation it affects arterial oxygen content and therefore oxygen delivery. By influencing how readily oxygen dissociates from hemoglobin it also affects venous oxygen content and the extraction of oxygen in tissues.
Affinity is not fixed. It can change in response to physiological conditions or structural differences in hemoglobin. These changes are described as shifts of the oxygen dissociation curve: a left shift indicates higher oxygen affinity, whereas a right shift indicates lower oxygen affinity.
Increasing affinity improves oxygen loading in the lung but makes oxygen more difficult to release in tissues. Decreasing affinity has the opposite effect, facilitating oxygen unloading but reducing the efficiency of oxygen loading.
Thus, a single change in hemoglobin affinity produces opposite effects on these two processes: oxygen loading in the lung and oxygen unloading in tissues. These opposing effects introduce a physiological trade-off between oxygen loading in the lung and oxygen unloading in tissues.
As we will see in the next section, shifts in hemoglobin affinity reveal this trade-off when the oxygen dissociation curve is examined across the range of oxygen tensions encountered in the lung and in peripheral tissues.
The curve reveals a trade-off
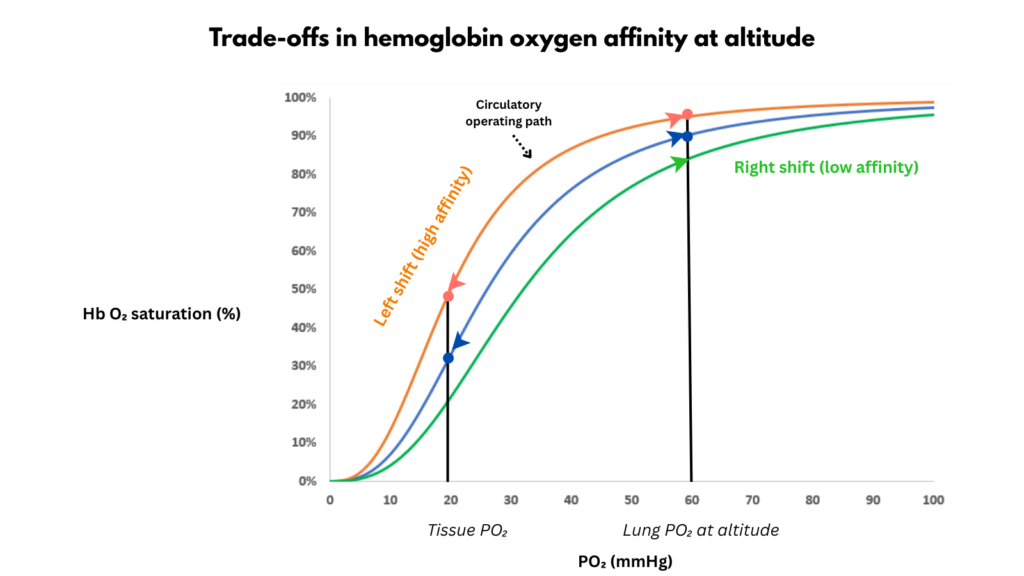
Hemoglobin affinity for oxygen can shift.
A left shift increases oxygen affinity.
A right shift decreases oxygen affinity.
Higher-affinity hemoglobin binds oxygen more tightly. This improves oxygen loading when lung PO₂ is low but makes oxygen more difficult to release in tissues.
Lower-affinity hemoglobin has the opposite effect: oxygen is released more readily in tissues, but loading in the lung becomes less efficient.
When these shifts are examined on the oxygen dissociation curve, they reveal a physiological trade-off between oxygen loading in the lung and oxygen unloading in tissues. A shift in either direction improves one side of this system while impairing the other.
The oxygen dissociation curve shows how shifts in hemoglobin affinity affect oxygen loading and unloading, but it does not determine which affinity is physiologically optimal.
At sea level, where oxygen is abundant, hemoglobin with either higher or lower oxygen affinity may function adequately. But when oxygen becomes scarce, as it does at high altitude, the balance between oxygen loading in the lung and oxygen unloading in tissues becomes a central physiological question.
Which shift in hemoglobin affinity is most adaptive under these conditions — a left shift that favors oxygen loading, or a right shift that favors oxygen unloading?
To answer that question, we must look beyond the curve to evidence from physiology, evolution, and human natural experiments.2
| Shift of oxygen dissociation curve | Hemoglobin affinity | Oxygen loading in lung (low PO₂ conditions) | Oxygen unloading in tissues |
|---|---|---|---|
| Left shift | Higher | Improved | Impaired |
| Right shift | Lower | Impaired | Improved |

When oxygen becomes scarce
At high altitude, atmospheric pressure falls and the partial pressure of oxygen in the lung declines. Under these conditions, hemoglobin molecules encounter oxygen less frequently in the alveoli, and arterial oxygen saturation begins to fall.
A left shift of the oxygen dissociation curve could help preserve oxygen loading in the lung. But increasing hemoglobin affinity also makes oxygen more difficult to release in tissues. A right shift would have the opposite effect, facilitating oxygen unloading but further impairing oxygen loading in the lung.
Simply inspecting the curve does not tell us which strategy is advantageous.
The curve shows the trade-off. It does not tell us which side of the trade-off matters most.
Human acclimatization to altitude
Humans ascending to high altitude undergo several physiological adjustments.
Ventilation increases within minutes, raising alveolar oxygen tension and partially restoring arterial oxygen saturation. Over the next several days, concentrations of 2,3-bisphosphoglycerate (2,3-BPG) in red blood cells increase, shifting the oxygen dissociation curve slightly to the right and facilitating oxygen release in tissues. Over longer periods, erythropoietin stimulates increased red blood cell production, raising hemoglobin concentration and improving oxygen transport.
These adjustments illustrate that humans respond to hypoxia through multiple mechanisms within the oxygen transport system.
In other words, hemoglobin affinity is only one lever within a larger physiological network that also includes ventilation, cardiac output, hemoglobin concentration, and microvascular oxygen extraction.
Human acclimatization therefore represents a system-level compromise, balancing oxygen loading, oxygen unloading, ventilation, blood flow, and red cell mass. The rise in 2,3-BPG likely reflects a response optimized for moderate hypoxia and normal tissue oxygen demand, where facilitating oxygen unloading remains advantageous, even if that strategy may become less optimal under more extreme oxygen limitation.
Yet these adjustments still do not resolve the central question posed by the oxygen dissociation curve:
When oxygen becomes scarce, is it more advantageous to improve oxygen loading in the lung or oxygen unloading in tissues?
When loading becomes the limiting step
Situations arise in biology in which oxygen loading rather than oxygen unloading becomes the dominant constraint.
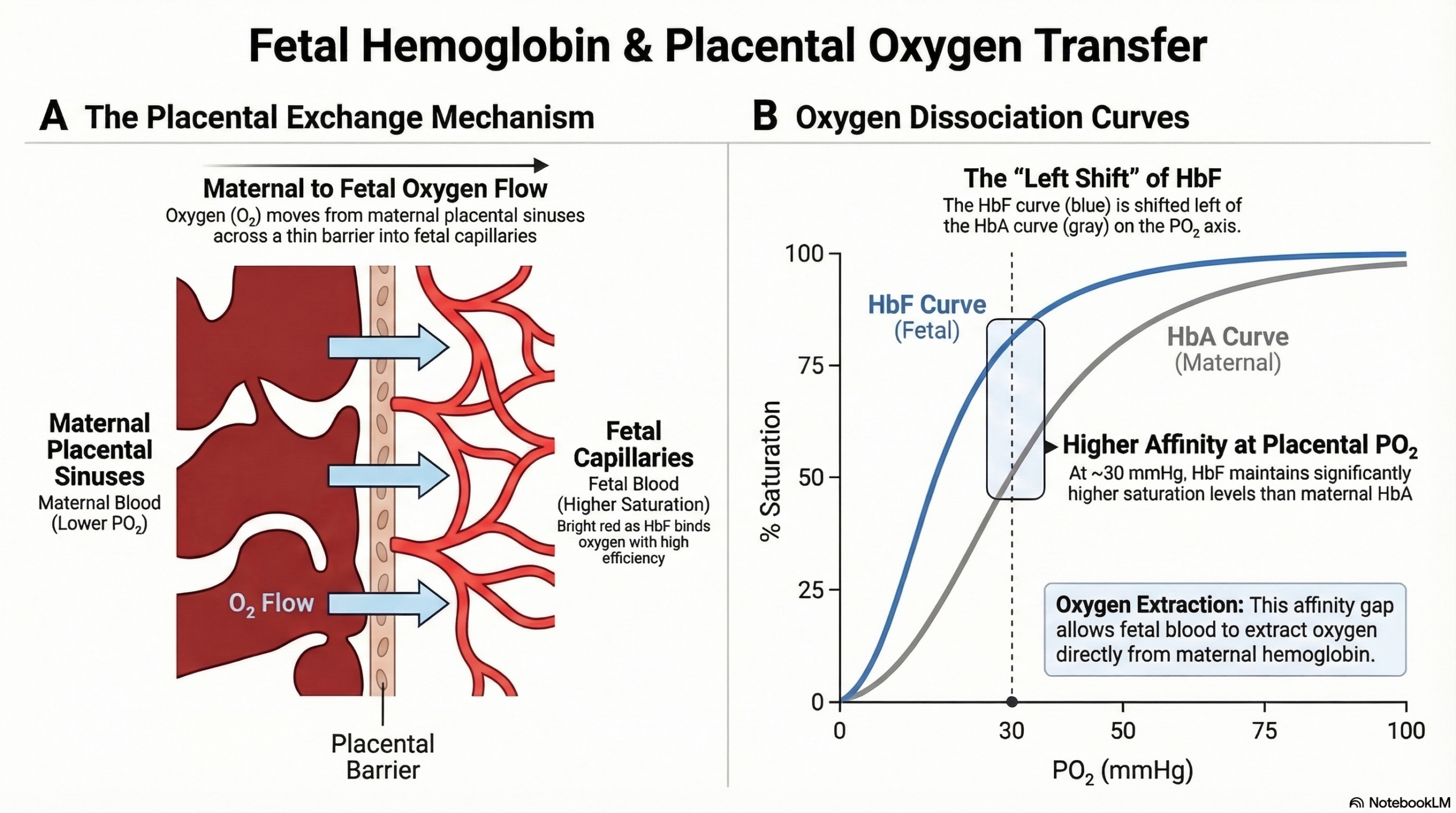
One example occurs during fetal life. Oxygen must diffuse from maternal blood across the placenta into the fetal circulation. Because the partial pressure of oxygen in maternal venous blood is relatively low, the fetus faces a challenging environment for oxygen uptake.
The fetus solves this problem with a specialized form of hemoglobin.
Fetal hemoglobin (HbF) has a higher affinity for oxygen than adult hemoglobin (HbA), shifting the oxygen dissociation curve to the left. This increased affinity allows fetal blood to bind oxygen more effectively at the relatively low oxygen tensions present in the placental circulation.
Fetal hemoglobin therefore represents a physiological solution to a setting in which oxygen loading is the dominant constraint.
Physiological evidence: fetal hemoglobin improves oxygen loading
Several physiological observations demonstrate how fetal hemoglobin facilitates oxygen transfer across the placenta.
Low placental oxygen tension
The partial pressure of oxygen in the intervillous space of the placenta is typically about 25–35 mmHg, far lower than the PO₂ in the adult lung.3
Higher oxygen affinity of HbF
Fetal hemoglobin has a P50 of ~19 mmHg, compared with ~26–27 mmHg for adult hemoglobin.4
This difference shifts the fetal oxygen dissociation curve to the left.
Greater saturation at the same PO₂
At a placental PO₂ of ~30 mmHg, fetal hemoglobin achieves a substantially higher oxygen saturation than adult hemoglobin.
Molecular mechanism
HbF interacts less strongly with 2,3-bisphosphoglycerate (2,3-BPG) than HbA. Reduced 2,3-BPG binding increases oxygen affinity.
Together these features allow fetal blood to extract oxygen from maternal blood despite the relatively low oxygen tensions present in the placental circulation.
Note: Illustrations in this box and the boxes that follow were created by the author using AI-assisted tools for educational purposes.
Evolution as a natural experiment
Nature has repeatedly conducted the altitude experiment.
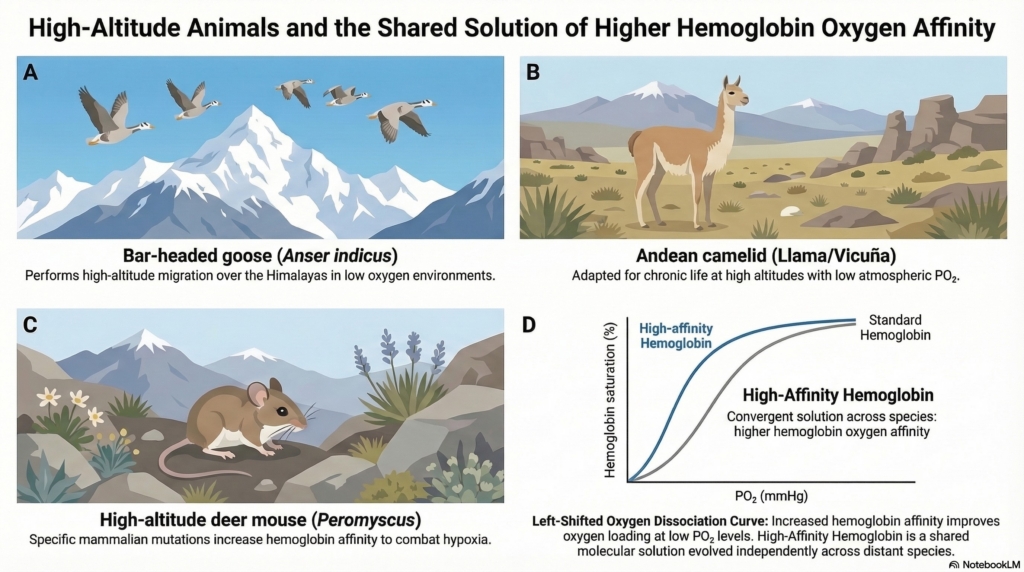
Many animals that live at high altitude possess hemoglobin variants with higher oxygen affinity, shifting the oxygen dissociation curve to the left. This pattern has evolved independently in several species, including bar-headed geese that migrate over the Himalayas and mammals such as llamas and other Andean camelids, and deer mice.
In these species, hemoglobin binds oxygen more readily at the lower oxygen pressures encountered at altitude. These evolutionary adaptations suggest that when oxygen becomes scarce, preserving oxygen loading in the lung becomes a dominant physiological constraint.
Evolution therefore offers one natural experiment in how organisms solve the altitude problem.
Natural experiments in evolution
How high-altitude animals solve the oxygen problem
Animals that live at high altitude face chronically reduced oxygen availability. Many species have evolved hemoglobins with higher oxygen affinity, allowing more effective oxygen loading at the lower PO₂ values encountered at altitude.
Examples include:
Bar-headed goose (Anser indicus)
This species migrates over the Himalayas at altitudes exceeding 7,000 meters, where oxygen levels are extremely low. Amino-acid substitutions in the hemoglobin α-chain reduce structural constraints within hemoglobin, increasing oxygen affinity and facilitating oxygen uptake in hypoxic air.5
Andean camelids (llama, alpaca, vicuña)
Camelids that inhabit the Andes possess hemoglobins with intrinsically high oxygen affinity and reduced sensitivity to 2,3-bisphosphoglycerate (2,3-BPG). These properties allow effective oxygen loading despite the low atmospheric oxygen pressures found at high altitude.7
These evolutionary solutions illustrate a recurring pattern:
Across diverse species, evolution repeatedly solves the altitude problem by increasing hemoglobin affinity and preserving oxygen loading in the lung.
Hemoglobin adaptations in high-altitude animals
The molecular mechanisms vary across species, but the physiological outcome is strikingly consistent.
| Species | Habitat | Hemoglobin adaptation | Physiological effect |
|---|---|---|---|
| Bar-headed goose (Anser indicus) | Himalayan migrations up to ~7,000–9,000 m | Amino-acid substitutions in the α-chain reduce constraints within the hemoglobin molecule | Increased oxygen affinity, improving oxygen loading in severely hypoxic air |
| Andean camelids (llama, alpaca, vicuña) | Andes (~3,500–5,000 m) | Hemoglobin with intrinsically high oxygen affinity and reduced sensitivity to 2,3-BPG | Improved oxygen loading at low atmospheric oxygen pressure |
| High-altitude deer mouse (Peromyscus maniculatus) | North American mountain ranges | Multiple hemoglobin mutations affecting both α and β chains | Increased oxygen affinity and improved aerobic performance in hypoxia |
A human natural experiment
Nature has also conducted a similar experiment in humans.
Rare genetic variants can produce hemoglobins with unusually high oxygen affinity. Individuals who carry these variants often develop erythrocytosis, as their bodies respond to reduced tissue oxygen delivery by increasing red blood cell production.
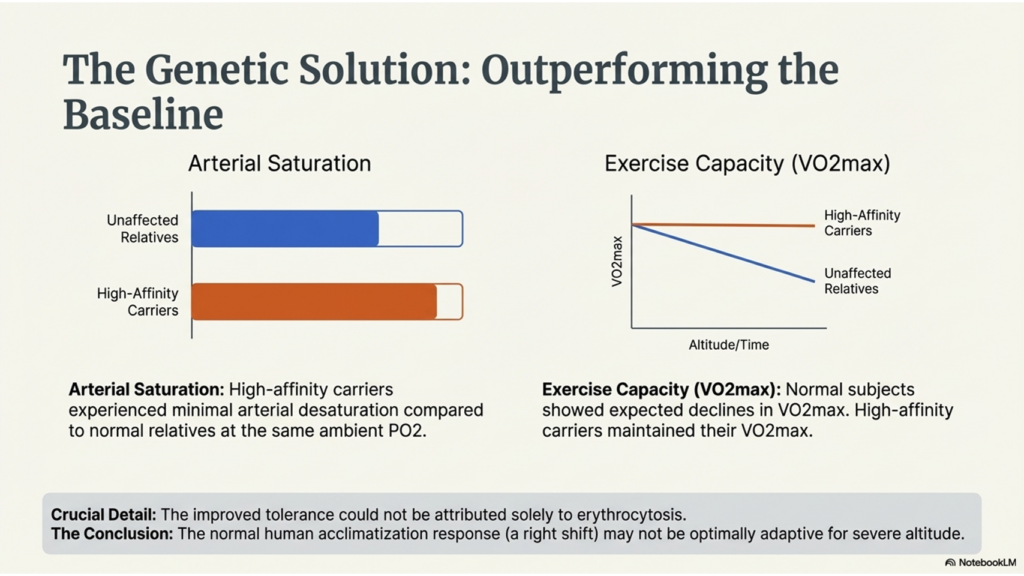
In a study by Hebbel and colleagues, a family carrying a high-affinity hemoglobin variant was examined during ascent to high altitude.8 Compared with unaffected relatives, individuals with the high-affinity hemoglobin tolerated altitude better and maintained higher arterial oxygen saturation.
In effect, these individuals behaved physiologically like high-altitude mammals such as llamas and other Andean camelids, whose hemoglobins bind oxygen more tightly.
The study provided a rare glimpse of what happens when the oxygen dissociation curve is shifted to the left in humans. Under conditions where oxygen becomes scarce, increased hemoglobin affinity improved oxygen loading in the lung, even though it could potentially impair oxygen unloading in tissues.
This human “natural experiment” echoed the lesson suggested by evolutionary biology: when oxygen availability falls, preserving oxygen loading becomes a dominant constraint.
Evidence from a human natural experiment
High-affinity hemoglobin and altitude tolerance
A family carrying a high-affinity hemoglobin variant was studied during ascent to high altitude.9 The mutation shifts the oxygen dissociation curve to the left, increasing hemoglobin affinity for oxygen.
Key observations included:
Higher arterial oxygen saturation (predicted)
Based on the measured oxygen affinity of the mutant hemoglobin, individuals with the high-affinity variant were predicted to experience minimal arterial desaturation at altitude, whereas relatives with normal hemoglobin would fall to lower arterial oxygen saturation at the same ambient PO₂.
Preserved exercise capacity
At altitude, normal subjects showed the expected decline in maximal oxygen consumption (VO₂max), whereas individuals with the high-affinity hemoglobin maintained their VO₂max, indicating preserved physiological performance under hypoxic conditions. HEBBEL
Effect not explained by erythrocytosis
Although high-affinity hemoglobin variants are often associated with erythrocytosis, the improved altitude tolerance observed in this study could not be attributed solely to increased hemoglobin concentration.
Mechanistic implication
The left-shifted oxygen–hemoglobin dissociation curve enhances oxygen loading in the lungs when ambient PO₂ is reduced. Under these conditions, preserving oxygen loading may be more physiologically important than facilitating tissue unloading.
Together, these findings provide rare human evidence that increased hemoglobin oxygen affinity can be advantageous when environmental oxygen availability is limited. They support the hypothesis that the normal human response to altitude—a modest right shift of the oxygen–hemoglobin dissociation curve—may not be optimally adaptive.
The lesson of the curve
The oxygen dissociation curve reveals a fundamental trade-off between oxygen loading in the lung and oxygen unloading in tissues.
But the curve itself does not dictate which side of that trade-off is optimal.
For that answer, we must look beyond the curve to physiology, experiment, and evolution.
The recurring lesson is not simply that higher hemoglobin affinity can be advantageous when oxygen becomes scarce. It is that physiological curves can define a problem without resolving it.
They display constraints.
They reveal tensions.
They map possible solutions.
But they do not tell us which solution biology will choose.
That answer emerges only when the curve is placed back into the living system — in organisms, in evolution, and ultimately in patients.

