Metaphor, Meaning, and the Limits of Naming in Hematology
Note: The video and audio linked above were generated with the assistance of AI. Clinical accuracy has been reviewed, but no AI-generated content can be guaranteed to be fully error-free.
Look through a microscope at a peripheral smear and the first thing you encounter is not biology, but language.
Target cells.
Teardrops.
Spurs.
Burrs.
Sickles.
Before we interpret physiology, we name what we see. In doing so, we decide, often unconsciously, what counts as a feature and what fades into the background. These names feel natural, almost inevitable. Yet they are neither. They are metaphors, and like all metaphors, they do more than describe. They shape how we perceive, how we reason, and sometimes how we misunderstand.
This essay traces how those words arise, how they evolve, and how they quietly guide both diagnosis and thought.
To understand how hematologists see, one must first understand how hematologists speak.
Seeing Through Borrowed Worlds
Every morphologic metaphor begins as an act of comparison.
At first, the language functions as analogy. We say that a cell looks like a target or resembles a teardrop. An analogy makes similarity explicit. It helps us describe what we see.
With time, the analogy transforms into metaphor. The language shifts from “looks like” to “is.” The cell becomes a target cell, a teardrop cell, a sickle cell. The comparison is no longer merely descriptive. It becomes a name. In time, such metaphors may become what linguists call dead metaphors: terms whose original imagery fades as they settle into technical vocabulary.
In linguistic terms, the red cell is the target domain, the object we seek to understand. The familiar object we invoke is the source domain, the world from which we import meaning. The mapping runs from the known to the unknown.
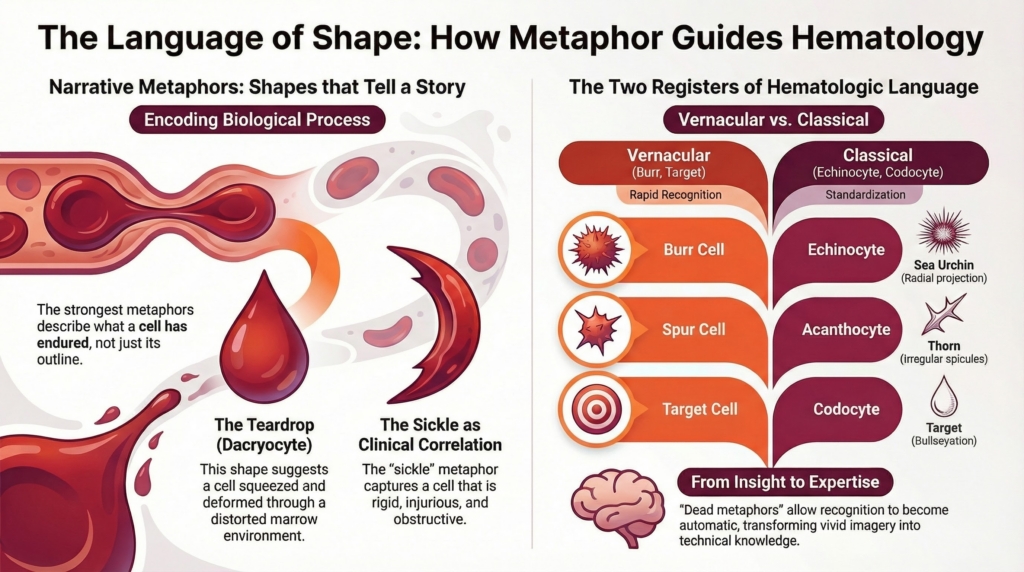
Consider the target cell. Its central disk and peripheral rim mirror the concentric zones of a bullseye, directing our attention to the relationship between center and edge. The metaphor does not simply label a shape. It guides perception toward a particular pattern.
In this sense, morphology is not merely visual. It is interpretive. We do not simply see cells. We see them through borrowed worlds.
When Metaphors Do More Than Describe Shape
Not all metaphors are equally powerful.
Some function only as visual shorthand. A triangular fragment or an ovalocyte tells us little beyond outline. The comparison is purely geometric.
The most enduring metaphors do something more. They capture not only how a cell looks, but something about what has happened to it.
A sickle is not merely curved. It is rigid, sharp, and capable of cutting. These associations mirror the biology of the sickled red cell: stiff, injurious, and obstructive. First described in 1910, the sickle cell became one of the earliest clinico-pathologic correlations in hematology. Over time, the metaphor extended far beyond the microscope, becoming the name of a disease, a genetic trait, and a source of social meaning.
A teardrop suggests deformation under pressure, as though the cell has been pulled or squeezed. In marrow fibrosis, that is precisely its story: cells forced through a distorted marrow environment.
In these cases, the metaphor does not simply describe form. It encodes process. The strongest morphologic metaphors are narrative. They help us understand not just what a cell is, but what it has endured.
From Microscope to Mind
Morphologic metaphors are born in vision but rarely remain alive.
At first, language functions as analogy. A cell looks like a target or resembles a teardrop. The comparison is explicit. The source and the cell are held together in view.
But the tension does not persist. In morphology, it is not meant to. What begins as metaphor quickly settles into terminology. “Target cell” ceases to be an active comparison and becomes the name of a category. Few clinicians consciously picture a shooting target; fewer still visualize an agricultural sickle when speaking of sickle cells. The metaphor has died.
Its death is not loss but stabilization. What remains is the structure the image once illuminated: center and rim, curvature and rigidity, symmetry and distortion. The imagery recedes, but the pattern endures.
In poetry, a metaphor lives by sustaining tension between source and target. In morphology, expertise depends on the opposite. The tension collapses so that recognition can become automatic. Live metaphor creates insight. Dead metaphor creates expertise.
The microscope is where metaphors are born. The mind is where they settle into knowledge.
Two Registers of Metaphor
Hematology does not speak in metaphor and non-metaphor. It speaks in two registers of metaphor.
One is vernacular and immediate: burr cells, spur cells, teardrops, targets. These names are vivid, culturally grounded, and cognitively efficient. They facilitate rapid recognition and bedside teaching.
The other is classical and standardized: echinocytes, acanthocytes, dacryocytes, codocytes. These Greek-derived terms appear purely technical, yet they too are rooted in metaphor. “Echinocyte” evokes a sea urchin, “acanthocyte” a thorn, “dacryocyte” a tear. They are metaphors translated into the formal language of scientific classification.
At times the two registers illuminate different aspects of the same cell. “Echinocyte” suggests a three-dimensional sea urchin, emphasizing radial projection, while “burr cell” evokes a flatter, tactile object. Each directs attention differently.
This distinction becomes especially visible when the same morphology is named differently in different clinical contexts. The irregular projections of an acanthocyte are identical to those of a spur cell. Yet in alcoholic cirrhosis the vernacular name persists, while in other disorders the classical term dominates. The morphology does not change. The linguistic register does. The difference reflects not structure, but context.
The difference between these systems is not the presence or absence of metaphor, but their function. Vernacular metaphors help us see. Classical metaphors help us standardize.
The Quiet Death of Metaphors
Over time, many metaphors lose their imagery while their words persist. These are not obsolete metaphors, but dead ones.
Few clinicians consciously picture a riding spur when identifying a spur cell or a plant burr when describing a burr cell. The words remain in active use, yet their visual origins have faded from awareness. They function now as technical labels rather than comparisons.
This process differs from historical obsolescence. Some early metaphors, drawn from the everyday objects of their time, simply fell out of use. Others survived but became so familiar that their imagery disappeared. The former were abandoned; the latter died.
Most morphologic metaphors in hematology belong to this second category. They are linguistic fossils, preserving earlier ways of seeing long after the visual comparison has faded.
Occasionally, a new metaphor enters the field before it has had time to settle. A recently described “fish cell,” reported in iron deficiency and thalassemia, is one such example. The form resembles a teardrop whose projection has split, evoking fins and a tail. Because the mapping has not yet settled into routine usage, the comparison remains active. One cannot hear “fish cell” without picturing a fish. The metaphor is still alive.
In time, it too may harden into terminology. For now, its imagery remains active.
The Aesthetics of Disease
Metaphors in hematology sometimes carry an aesthetic charge, but that charge is not about beauty or severity. It is about pattern.
When examining a smear with trainees, burr cells may appear orderly. Their projections are evenly spaced, relatively uniform in length, and symmetrically distributed. Spur cells, by contrast, look irregular. Their spicules vary in size, spacing, and orientation, giving the outline an uneven contour. The difference is not one of attractiveness, but of structural regularity.
The eye is exquisitely sensitive to symmetry and its disruption. Before a cell is named, it is registered as patterned or unpatterned, coherent or heterogeneous. What feels “wrong” is often the visual system detecting irregularity before the mind has articulated why.
This aesthetic response is therefore not separate from morphologic reasoning. It is an early stage of it. Recognition begins not with terminology but with pattern detection. The eye classifies before the mind names.
Where Naming Ends
The limits of metaphor become most visible in fragmentation.
In microangiopathic hemolysis, shear stress produces recurring fracture patterns. Helmet cells, triangular fragments, and crescents form a reproducible repertoire of shapes collectively recognized as schistocytes. Even though they arise from destruction, they follow predictable geometries that invite naming.
But when injury becomes diffuse, as in thermal damage or hemoglobin H disease, fragmentation produces irregular, non-repeating forms. No consistent pattern emerges. At this boundary, language retreats to the generic term “fragments.”
The distinction is therefore not between shape and destruction, but between pattern and variability. Even destructive processes can sustain rich vocabulary when they produce stable forms. Only when irregularity becomes complete does naming collapse.
Language follows pattern more readily than process.
Metaphors as Cultural Artifacts
Morphologic metaphors also reflect the world of those who coined them.
Early hematology literature reveals a remarkable abundance of analogies drawn from everyday life. Around the turn of the twentieth century, red cells were described as pears, balloons, saucers, and canoes. A decade later, observers invoked tennis rackets, tomahawk blades, dumbbells, and anvils.
These comparisons arose from a material world rich in familiar objects and crafts. A physician in 1910 might readily visualize an anvil or a belaying pin, objects embedded in daily life. Today, such metaphors feel obscure, not because the cells have changed, but because the world of the observer has. Strictly speaking, many of these are not dead metaphors but obsolete ones: terms that disappeared altogether rather than surviving with their imagery faded.
Morphologic language, like language itself, evolves with culture.
The Double Edge of Metaphor
Metaphors illuminate, but they also constrain.
By highlighting certain features, they inevitably obscure others. A trainee may quickly recognize a target cell yet struggle to explain how excess membrane relative to volume produces the bullseye appearance. The metaphor guides perception but may delay mechanistic understanding.
Every metaphor is therefore both a window and a filter. It clarifies what we see, even as it subtly shapes how we think.
Seeing, Naming, and Understanding
At the microscope, we do more than observe cells. We translate them into stories drawn from the human world.
Metaphors allow us to bridge scales, transform perception into communication, and encode complex biological processes into memorable language. Yet they also reveal the limits of human understanding.
We can name what is stable. We can compare what is recognizable. But where biological variability exceeds the limits of pattern, language falters.
Each smear, therefore, is not only a field of cells. It is also a field of language, one that illuminates how we see and where our ability to name reaches its limits.
