How judgment, not algorithms, shapes treatment in cold agglutinin disease
Note: The video and audio linked above were generated with the assistance of AI. Clinical accuracy has been reviewed, but no AI-generated content can be guaranteed to be fully error-free.
Why this overview exists
Cold agglutinin disease (CAD) is unusual among hematologic disorders in that effective therapies exist, yet no single treatment strategy is universally correct. This is not because the biology is unclear, but because the clinical questions vary. CAD is a clonal B-cell disorder in which monoclonal IgM antibodies activate the classical complement pathway, driving predominantly extravascular hemolysis, with variable intravascular contribution. That biology is well established.2
What remains uncertain, and inherently contextual, is how best to intervene in a given patient, at a given moment. Some patients need hemolysis stopped quickly, others seek durable disease modification, and many will require both at different times and in different sequences. Understanding treatment in CAD therefore begins not with drugs, but with goals.3
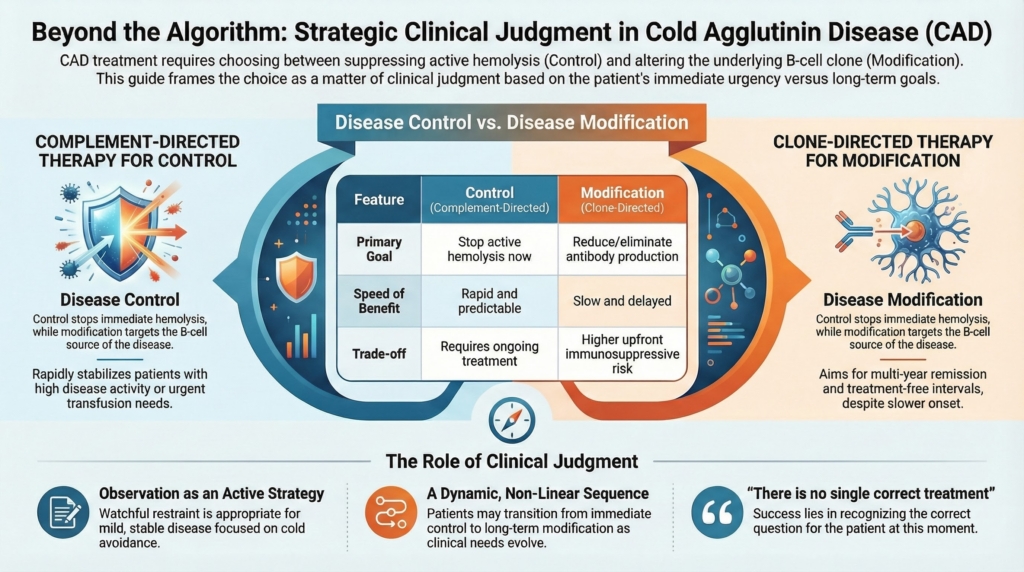
This overview frames CAD treatment strategy around two therapeutic priorities—rapid control of complement-mediated hemolysis and longer-term modification of the IgM-producing clone—while recognizing that clinical judgment determines how they are sequenced or combined.
Control, modification, and observation are not properties of the disease, but questions the clinician is asking of it.
The detailed mechanisms, evidence, and limitations of each approach are addressed in the companion essays on complement-directed and clone-directed therapy. Here, the task is orientation.
Two therapeutic goals, not one
All CAD therapies aim to reduce disease burden, but they do so by targeting different points along the disease pathway.4
Disease control focuses on suppressing active complement-mediated hemolysis.
Disease modification aims to alter the source of disease by reducing or eliminating pathogenic antibody production.
Neither goal is inherently superior. Each addresses a different clinical question, and in any given moment one may matter more than the other. A useful first step in CAD management is to clarify whether the immediate priority is control, modification, or observation.5
In many patients, both goals are partially relevant, and the task is not choosing one, but deciding which matters most right now.
Each clinical scenario that follows can be understood by asking the same question: what is the dominant priority right now—control, modification, or observation?
Control: stopping hemolysis now
Control-oriented therapy is concerned with immediacy and predictability. It asks:6
- Is hemolysis active and clinically consequential?
- Is anemia worsening, symptomatic, or transfusion-dependent?
- Is rapid stabilization required because of infection, surgery, comorbidity, or physiologic stress?
Proximal classical-pathway inhibition is particularly well suited to this goal. By interrupting the classical complement pathway upstream of C3, C1s inhibition suppresses the dominant effector mechanism in CAD and produces rapid, reproducible improvements in hemolysis and hemoglobin.7
When control is the dominant goal
Control strategies are particularly valuable when disease activity is high, tempo is fast or unstable, and the cost of waiting is unacceptable. They prioritize immediacy and predictability over durability, aiming to stabilize hemolysis and prevent near-term clinical consequences.
Therapies that act directly on the effector phase of disease, most notably complement inhibition, are therefore well suited to this goal. Because complement inhibition does not eliminate the underlying IgM-producing clone, continued treatment is generally required to maintain suppression of hemolysis, but that limitation is acceptable when the clinical problem is urgent stabilization rather than long-term disease redesign.8
Modification: changing the disease over time
Modification-oriented therapy addresses a different set of questions:9
- Can antibody production be reduced or suppressed?
- Is the disease tempo slow enough to tolerate delayed benefit?
- Is a finite course of therapy preferred over ongoing treatment?
- Can the patient tolerate immunosuppression?
Clone-directed strategies aim to suppress or eliminate the B-cell population responsible for IgM production, using rituximab-based regimens alone or in combination. Modification becomes the dominant goal when hemolysis is stable enough to tolerate delay, but the long-term disease trajectory or treatment burden argues against indefinite control-oriented therapy. The effects of clone-directed strategies are slower and less predictable in the short term, but they offer the possibility of treatment-free intervals and longer remissions.
When modification is the dominant goal
Modification strategies are most appealing when disease tempo is slower, hemolysis is stable or modest, and long-term trajectory outweighs short-term urgency. These approaches trade immediacy for durability, accepting higher upfront immunosuppressive risk for the potential of multi-year remission without continuous therapy.
Observation as an active strategy
Not all CAD requires immediate intervention. Observation, when chosen deliberately, is a legitimate and often appropriate strategy. Patients with mild anemia, limited symptoms, or stable disease trajectories may be best served by watchful restraint, with attention to cold avoidance, trigger management, supportive care, and periodic reassessment. Observation is not neglect.10
The decision to treat, and the decision not to treat, are both acts of judgment.
Why this is not an either–or decision
In practice, many patients will require both strategies over time: control to stabilize disease expression, and modification to reduce dependence on ongoing effector-phase therapy. Control and modification are often presented as competing strategies; in practice, they are better understood as complementary tools, applied at different moments along a disease course. The correct sequence is not fixed. It evolves.11
Where judgment enters
Guidelines and trials inform what therapies can do, but they do not determine what should be done for a given patient. Judgment is required to weigh urgency versus durability, predictability versus treatment burden, short-term control versus long-term strategy, and laboratory improvement versus lived experience.
In practice, these decisions may also be shaped by access, cost, and drug availability, which can constrain options independent of clinical reasoning.
For example, a patient with only moderate anemia by laboratory criteria may experience disabling fatigue or cold intolerance that justifies active control, while another with similar indices may feel well and reasonably prefer observation or delayed modification. This variability is not error; it is the nature of the disease.13
How to use the companion essays
This overview is intended to orient, not replace, the detailed therapy spokes. Complement-Directed Therapy explains how and why hemolysis can be rapidly suppressed, and where that strategy reaches its limits, drawing on data from C1s inhibition trials and real-world use of sutimlimab. Clone-Directed Therapy explores disease modification, durability, and the trade-offs of immunosuppression, using evidence from rituximab monotherapy and combination regimens. Guidelines, Evidence, and Judgment addresses why neither evidence nor guidance can substitute for clinical reasoning in CAD, summarizing international consensus and its gray zones. Read together with the Sequencing essay, these pieces reflect the reality of practice: treatment in CAD is not algorithmic. It is strategic.
A final principle
There is no single correct treatment for cold agglutinin disease; there is only a correct question for this patient, at this moment. Mastery lies in recognizing whether the problem is one of control, modification, or watchful restraint, and in revisiting that judgment as the disease, and the patient, evolve.
Reflect and Apply
Think of a recent patient with CAD, or another chronic hematologic condition, in whom you felt uncertain about treatment.
Pause and ask yourself:
- what question were you actually trying to answer at that moment: control, modification, or observation?
- did you explicitly name that goal before choosing therapy?
- what factor most influenced your decision: urgency, trajectory, symptoms, or patient priorities?
- if another clinician chose differently, would that represent error, or different weighting?
Now imagine seeing the same patient again today.
Would your dominant goal be the same?
If not, what changed: the disease, the patient, or your interpretation?
The goal is not agreement. The goal is to make your weighting visible.
Test your thinking
A short, judgment-focused quiz that complements this overview by testing treatment strategy decisions in cold agglutinin disease.
