Active care beyond disease-directed therapy
Note: The video and audio linked above were generated with the assistance of AI. Clinical accuracy has been reviewed, but no AI-generated content can be guaranteed to be fully error-free.
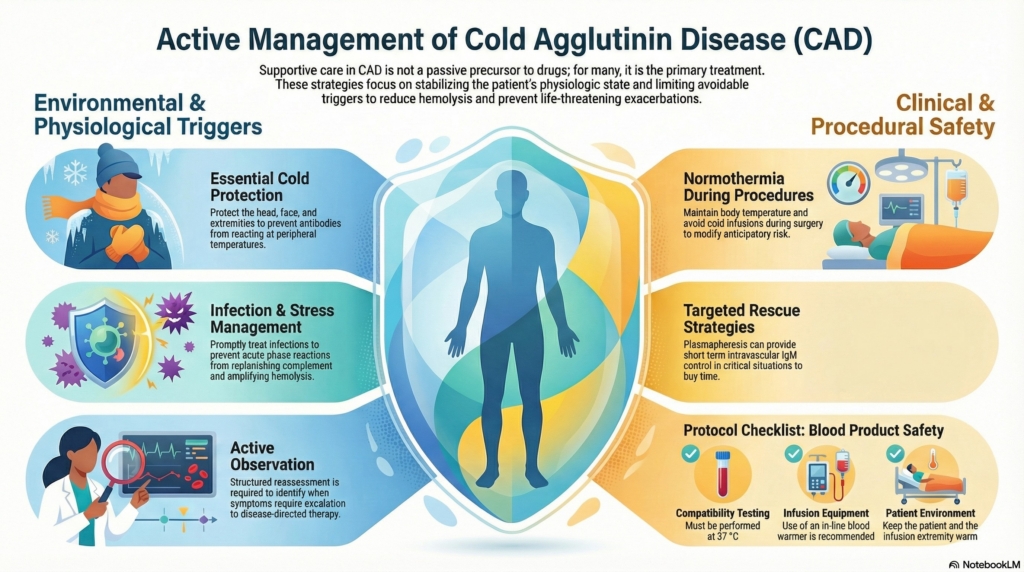
Why this spoke matters
Supportive care is not ancillary in cold agglutinin disease; historically, counseling and non-pharmacologic measures were considered the mainstay of management because effective drug therapy was limited.1
Not all patients require pharmacologic treatment, and some may be managed without disease-directed therapy, making supportive care central to clinical management. In this sense, supportive care is not what precedes treatment. For some patients, supportive care may be the main management strategy.2
The unifying idea is simple: supportive care modifies risk and expression of disease, even when it does not alter its cause.
What supportive care is trying to accomplish
Supportive measures aim to reduce hemolysis, mitigate symptoms, and prevent exacerbations in a disorder characterized by chronic complement-mediated red-cell destruction and cold-induced circulatory symptoms.3
This reflects the dual pathophysiology of CAD: complement-driven hemolysis and non–complement-mediated agglutination, which contribute differently to anemia and circulatory symptoms.4
Because disease expression varies widely, management focuses not only on altering the disease process but also on stabilizing the patient’s physiologic state and reducing avoidable triggers. These interventions do not alter the clonal biology or complement pathway; instead, they limit precipitating conditions and reduce downstream clinical consequences.
Supportive care in CAD: intervention → trigger → clinical effect
| Intervention | Trigger or physiologic context | Clinical effect |
|---|---|---|
| Thermal protection | Peripheral cooling, IgM binding in cooler vascular beds | Reduces circulatory symptoms, may reduce hemolytic triggering |
| Infection management | Acute-phase complement replenishment | Limits exacerbation of hemolysis |
| Transfusion precautions (warming, 37°C testing) | Iatrogenic cold exposure, in vitro agglutination | Enables safe transfusion, avoids acute worsening |
| Perioperative normothermia | Hypothermia during procedures | Reduces risk of peri-procedural hemolysis |
| Structured monitoring | Disease fluctuation over time | Detects progression and informs escalation |
| Folic acid supplementation | Increased erythropoietic demand from chronic hemolysis | Supports red-cell production (evidence limited) |
Cold protection: necessary but not sufficient
Physiologic cooling of peripheral blood may be sufficient to provoke complement-mediated hemolysis or cold-induced circulatory symptoms in patients whose antibodies react at relatively high temperatures.5
Avoidance of cold exposure is widely recommended, particularly protection of the head, face, and extremities.6
Patients should therefore:
- avoid cold environments when possible
- use warm clothing
- minimize peripheral cooling
These measures may alleviate symptoms and reduce the risk of exacerbations, although evidence for magnitude of benefit is limited.7
Even with careful cold avoidance, symptomatic disease can persist, and severe manifestations can occur even in warm climates.8 Persistent hemolysis despite reasonable protective measures does not imply poor adherence or mild disease. It reflects biologic variability rather than behavioral failure.
Cold avoidance recommendations can impose real lifestyle, occupational, and psychological burdens, particularly in colder climates. The goal is risk reduction, not social isolation.
Transfusion support
Transfusion can be safely administered when clinically indicated, provided appropriate precautions are followed.9
Important principles include:10
- compatibility testing should be performed at 37°C, which usually makes compatibility easier to establish than in warm autoimmune hemolytic anemia
- the patient and infusion extremity should be kept warm
- use of an in-line blood warmer is recommended
Failure to maintain warming measures can precipitate hemolytic complications.11
Transfusion remains supportive therapy and can be used safely when clinically indicated, including for urgent correction of severe anemia. When patients become transfusion-dependent, this reflects disease burden and severity rather than failure of supportive care.
Acute rescue strategies
In critical situations requiring rapid control of hemolysis, plasmapheresis can be used as a short-term intervention because IgM antibodies are largely intravascular.12
The effect is brief, and evidence is limited to case reports and small series. Its value lies in buying time, not in altering disease trajectory.13
Infection and physiologic stress
Exacerbations of hemolysis may occur during febrile infections, trauma, or major surgery.14
This phenomenon is thought to reflect complement dynamics:15
- steady-state patients often show complement consumption, commonly with low C4
- acute phase responses can increase hepatic production of complement proteins, including C3 and C4, replenishing substrate for classical pathway activation
- increased complement availability may amplify complement-mediated hemolysis
The key point is that acute worsening does not necessarily imply clonal progression; it may reflect increased complement availability during inflammation or physiologic stress.16
Prompt treatment of infection is recommended because intercurrent infection can precipitate worsening hemolysis.17
Procedural and perioperative planning
Patients with CAD undergoing procedures require specific precautions. Hypothermic procedures can provoke clinically significant hemolytic complications in susceptible patients, especially when cold agglutinins have high thermal amplitude. In these settings, supportive care functions as anticipatory risk control, not adjunctive therapy.18
Recommended principles include:19
- maintain normothermia during procedures
- avoid cold infusions
- obtain hematology evaluation before major surgery
Anticipatory planning is essential because perioperative cooling, unwarmed fluids, or hypothermic procedural conditions can shift a stable patient into active hemolysis.
Monitoring during observation
Some patients with CAD do not require immediate pharmacologic therapy, but this does not eliminate the need for active clinical follow-up.20
Patients managed without immediate pharmacologic therapy still require periodic reassessment for anemia, hemolysis, symptoms, and evolving treatment indications.21 Longitudinal cohort data show that anemia severity in CAD fluctuates over time, with many patients experiencing recurrent severe anemia events despite therapy, reinforcing that disease status cannot be inferred from a single time point.22
What supportive care cannot do
Supportive measures reduce exposure to triggers and limit downstream consequences, but they do not eliminate the monoclonal IgM-producing clone or reliably prevent complement-mediated hemolysis once antibody binding has occurred.
Disease-directed therapy targets one of two major biologic axes: the B-cell clone responsible for pathogenic IgM production, or the complement pathway responsible for red-cell destruction.
Clone-directed therapy aims to reduce antibody production and may provide more durable disease control, but responses are often delayed.23
Complement-directed therapy can rapidly control hemolysis by interrupting classical pathway activation, but it does not eliminate the underlying antibody-producing clone.24
Escalation decisions arise from convergence of symptoms, trajectory, functional impact, treatment burden, and patient priorities rather than from a single number.
The practical question is not only whether hemolysis is present, but which layer of the disease requires intervention: exposure, complement activation, antibody production, or some combination of these.
Explicit principle
Supportive care in cold agglutinin disease is active management, not therapeutic passivity.
Key Points
- supportive care is active treatment — it modifies risk, triggers, and physiologic stress even when no drugs are used
- not all CAD requires pharmacologic therapy — some patients are best managed with monitoring and preventive measures alone
- thermal protection, infection control, and transfusion precautions matter clinically because they reduce exposure to triggers and procedure-related aggravation, not because they alter the clone
- observation requires structure — follow-up is management, not absence of care
- escalation decisions are integrative — symptoms, trajectory, and functional impact matter more than a single laboratory value
- supportive care stabilizes disease but does not cure it — disease-directed therapy is required when burden exceeds physiologic tolerance
Test your thinking
A short, judgment-focused quiz on supportive care in cold agglutinin disease.
Evidence anchors
Key statements in this spoke are grounded in the following evidence:
- cold avoidance is recommended in CAD, although formal trial data demonstrating magnitude of benefit are limited
- in a cohort of 232 patients with primary CAD, approximately 24% required no pharmacologic therapy
- a substantial proportion of patients with CAD (up to 65%) require RBC transfusion at some point in their disease course
- transfusion precautions, including compatibility testing at 37 °C and use of blood warming, are supported by transfusion medicine guidance and case experience
- therapeutic plasma exchange produces temporary reductions in hemolysis in CAD but does not provide sustained disease control
- perioperative normothermia reduces risk of hemolytic exacerbation in patients with clinically significant cold agglutinins
Sources supporting these statements: Blood 2020;136:480; Transfus Med Hemother 2015;42:294; J Clin Apher 2023;38:77; Hematology Am Soc Hematol Educ Program 2015;2015:454; Mullins M et al. Cold agglutinin disease burden: a longitudinal analysis of anemia, medications, transfusions, and health care utilization. Blood Adv. 2017.
Guideline perspective: How supportive care fits into CAD management
How supportive care fits into CAD management
Major professional societies and expert reviews emphasize:
- advising patients to avoid cold exposure, particularly of the head and extremities
- maintaining normothermia during procedures and avoiding cold infusions
- performing compatibility testing at 37 °C and using blood warmers for transfusion
- treating infections promptly to reduce hemolytic exacerbation
- considering therapeutic plasma exchange as a short-term rescue strategy in severe or life-threatening hemolysis
- monitoring patients under observation with structured follow-up rather than passive watchful waiting
Guidelines position supportive care as foundational management across the disease spectrum. It is appropriate as sole therapy in mild disease and remains necessary even when disease-directed therapies are used.
Organizations referenced include:
- British Society for Haematology (BSH)
- American Society for Apheresis (ASFA)
- European expert consensus reviews
- ASH Education Program reviews
