Matching mechanism to moment
Note: The video and audio linked above were generated with the assistance of AI. Clinical accuracy has been reviewed, but no AI-generated content can be guaranteed to be fully error-free.
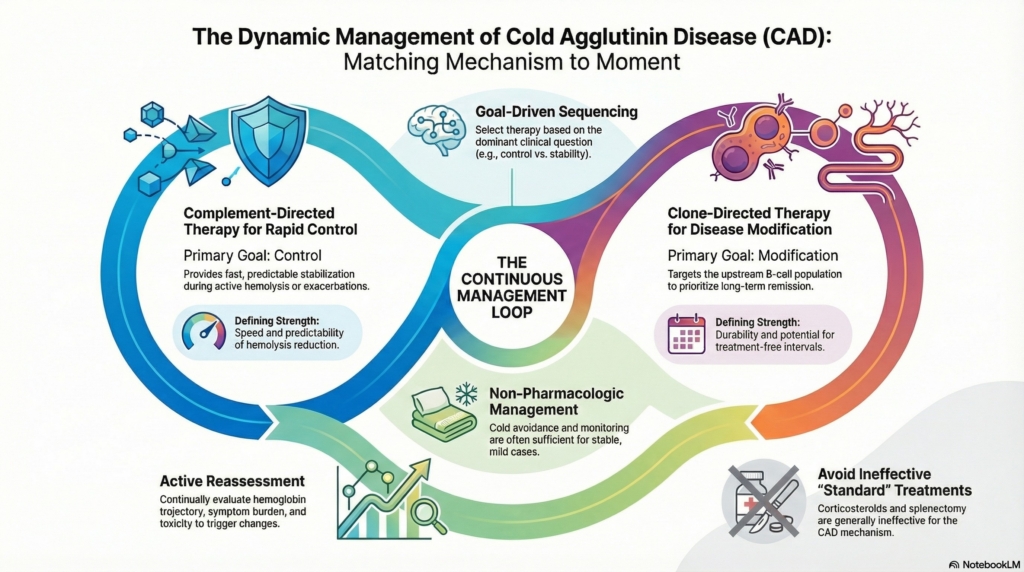
Why this spoke matters
Cold agglutinin disease is not managed with a single decision point but as a sequence of phases in which disease activity, mechanism, and patient priorities change. The task is not to climb a ladder but to keep matching mechanism to moment.1
This spoke is written for clinicians managing CAD longitudinally. Most patients move through multiple phases of care, not all of which require the same therapeutic strategy.2
Management is best conceptualized as a loop: goal setting, intervention, reassessment, and adjustment, rather than a one-way escalation pathway. Each phase of care can be read through the same lens: mechanism, tempo, and therapeutic goal.
CAD management is dynamic, not linear
CAD treatment is individualized rather than following a single uniform sequence.3
Instead, management involves repeated alignment between:4
- disease activity and tempo
- mechanism driving current expression
- therapeutic goals (control vs modification)
- patient tolerance for risk and chronic therapy
A strategy appropriate at one point in the disease course may become inappropriate later.5
Sequencing by clinical goal
Therapies in CAD answer different clinical questions, and sequencing follows the dominant question at a given moment.6
Typical goal-driven sequencing includes:7
- rapid control when hemolysis is active or symptomatic
- disease modification when stability allows a slower approach
- observation when disease impact is low
- supportive care throughout all phases
Supportive care is the constant background against which control, modification, and observation rotate.
This framing avoids false competition between therapies: the task is not to pick the “best drug,” but the best question to answer first.
When observation and supportive care are enough
Not all patients require drug therapy.8
Some individuals with mild anemia or limited symptoms can be managed with:9
- cold avoidance
- treatment of precipitating infections
- monitoring rather than intervention
Supportive strategies remain sufficient in selected patients, particularly when hemolysis is stable and symptoms are minimal.10 In such patients, the underlying clone may remain biologically stable for years, making treatment risk greater than disease burden.
Complement-directed therapy as rapid control
Complement activation is the central effector mechanism of hemolysis in CAD.11 Because of this, proximal complement inhibition can produce rapid improvement in hemolysis and anemia.12
Typical roles include:13
- first-line control for active hemolysis
- stabilization during exacerbations
- bridging while diagnostic clarification occurs
- protection during slower clone-directed therapy
Complement inhibition rapidly controls hemolysis but only while it continues, regulating activity without eliminating the upstream driver. Proximal C1s inhibition is the mechanistically appropriate target; terminal C5 blockade is largely ineffective in CAD.
Hemolysis commonly recurs after discontinuation of complement inhibition, underscoring its role as a control rather than curative strategy.
Clone-directed therapy as disease-modifying strategy
CAD is usually driven by a clonal B-cell population producing pathogenic IgM.14 In practice, clone-directed therapy is often introduced once hemolysis is stabilized, allowing time for slower disease modification to take effect.
Clone-directed therapy therefore targets the upstream disease mechanism rather than the downstream hemolysis.15
It is generally introduced when:16
- long-term remission is a priority
- disease is stable enough to tolerate delayed benefit
- the underlying lymphoproliferative disorder warrants treatment
- complement inhibition alone is insufficient
Rituximab monotherapy produces responses in roughly half of patients, though responses are often temporary.17
Combination regimens such as fludarabine-rituximab and bendamustine–rituximab produce higher response rates and longer remissions than rituximab monotherapy, at the cost of greater toxicity.18
Thus, regimen choice reflects a deliberate trade-off between durability and risk.19 The decision is rarely about efficacy alone; it is about how much durability the clinical situation justifies purchasing at the price of immunologic risk.
Therapies that generally should not be used
Some treatments effective in other autoimmune cytopenias are ineffective or inappropriate in CAD.20
These include:21
- corticosteroids
- splenectomy (in most cases)
Recognizing what not to use is part of expert sequencing. In typical primary CAD, corticosteroids and splenectomy are generally ineffective because IgM initiates classical-pathway activation, while hemolysis is mediated predominantly by complement deposition and largely hepatic extravascular clearance.
In mixed or atypical cases, treatment should follow the dominant mechanism rather than a fixed disease label.
Combination therapy: purpose, not redundancy
Complement-directed and clone-directed therapies act at different levels of disease biology and may be intentionally combined.22
Conceptual strategies include:23
- complement inhibition for immediate control while clone-directed therapy reduces antibody production
- overlap during transition phases
- stabilization during induction therapy
Combination therapy is goal-driven, not additive by default.24 Concurrent drugs do not necessarily represent combination strategy; sometimes they represent sequential logic executed safely.
For example, a patient with severe hemolysis may begin complement inhibition for immediate control while starting clone-directed therapy, then taper complement blockade once clonal suppression is achieved.
When combination therapy is not appropriate
Layering therapy is avoided when:25
- disease impact is mild
- risks of immunosuppression outweigh benefit
- patient preference favors minimal intervention
- control is adequate with a single strategy
Avoiding unnecessary escalation is as important as escalation itself. More therapy is not better therapy when it does not change outcomes that matter to the patient.26
Reassessment as an active management step
Reassessment is not passive monitoring.27 In chronic disease, reassessment is a therapeutic act because it prevents both overtreatment and undertreatment. This is the work that keeps the loop turning.
In CAD it determines whether the current strategy still matches disease biology and patient priorities.28
Key reassessment domains include:29
- hemoglobin trajectory
- biochemical hemolysis markers
- symptom burden
- exacerbation frequency
- transfusion need
- treatment toxicity
- patient goals
Reassessment informs whether to continue, escalate, de-escalate, or switch therapy. In longitudinal care, reassessment functions as a therapeutic intervention in its own right.30
Triggers for strategy change
Clinical signals that should prompt reconsideration include:31
- persistent symptomatic anemia
- breakthrough hemolysis
- increasing transfusion dependence
- unacceptable adverse effects
- evolving comorbidities
- progression of the underlying clone
Failure to respond to these signals leads to therapeutic inertia. Inertia is the default failure mode of chronic disease management because changing course requires both uncertainty tolerance and cognitive effort; the loop breaks when reassessment does not translate into action.32
De-escalation and stopping therapy
Not all treatment decisions involve escalation.33 From the outset, clinicians should distinguish therapies that are inherently time-limited from those that are intrinsically chronic, because this distinction shapes expectations and long-term planning.
De-escalation may be appropriate when:34
- disease activity diminishes
- treatment toxicity rises
- priorities shift
- burden outweighs benefit
In CAD, complement-directed therapy usually controls disease only while continued, whereas successful clone-directed therapy may allow treatment-free intervals.35
Understanding which therapies permit discontinuation is essential for long-term planning.36 This asymmetry between control therapies and modifying therapies is one of the central strategic facts of CAD management.
Long-term framing of CAD
For many patients, CAD behaves as a chronic but manageable disorder.
Expert management reframes care around:37
- anticipation rather than reaction
- aligning therapy with life context
- preventing avoidable exacerbations
- honest discussion of limits of cure
Sequencing and reassessment operationalize this philosophy.38 Longitudinal success depends less on any single decision than on repeatedly updating decisions as biology evolves.
Explicit principle
There is no single correct sequence in cold agglutinin disease.
Expert care lies in matching mechanism to moment and revisiting decisions as biology and priorities evolve.
Reassessment is not uncertainty — it is mastery.
Test your thinking
A short, judgment-focused quiz on sequencing in cold agglutinin disease.
Evidence anchors
Key statements in this spoke are grounded in the following evidence:
- complement inhibition produces rapid hemoglobin improvement
- rituximab monotherapy yields ~50% responses, often transient
- rituximab-based combinations increase response depth and duration but raise toxicity risk
- C5 blockade has limited efficacy in CAD because it does not prevent C3-mediated extravascular hemolysis
Sources supporting these statements: Berentsen S. How I treat cold agglutinin disease. Br J Haematol. 2011; Jäger U et al. Diagnosis and treatment of AIHA: international consensus. Front Immunol. 2020; British Society for Haematology. Guideline for autoimmune hemolytic anemia. Br J Haematol. 2016; Röth A et al. Sutimlimab in CAD. N Engl J Med. 2021; Berentsen S. Therapeutic advances in CAD. Hematology Am Soc Hematol Educ Program. 2016.
