Sustaining alignment as disease and life evolve
Note: The video and audio linked above were generated with the assistance of AI. Clinical accuracy has been reviewed, but no AI-generated content can be guaranteed to be fully error-free.
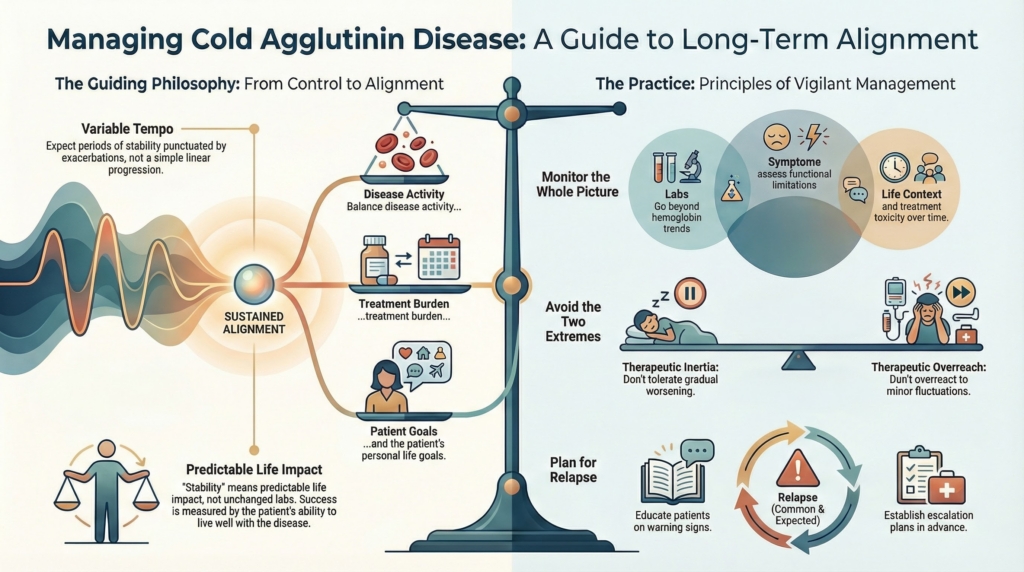
Why this spoke matters
Cold agglutinin disease is rarely a one-decision illness.1
Even when diagnosis is clear and an initial strategy is chosen, most patients live with CAD over years rather than months. Disease activity fluctuates, therapies change, risks accumulate, and priorities evolve.2
Expert care therefore depends not only on selecting therapy, but on knowing how to monitor, when to reassess, and how to adjust course without overreacting or drifting.3
This spoke addresses the central challenge of longitudinal CAD care: maintaining alignment between disease biology, treatment burden, and the patient’s lived reality over time.
CAD is a chronic condition with variable tempo
Most patients follow a relapsing or fluctuating course rather than a linear trajectory.4
Typical patterns include:
- stable periods interrupted by exacerbations
- symptoms that fluctuate independently of hemoglobin
- partial rather than complete responses
- relapse after treatment-free intervals
- evolving comorbidities and treatment tolerance
A distinct biologic feature of CAD is that the clonal B-cell disorder and hemolysis may evolve on different timelines. Clone progression may occur despite stable anemia, while hemolysis may worsen without clonal transformation. Long-term monitoring must therefore track both processes, not assume they move together.
Long-term management requires tolerance of imperfect but acceptable disease control. The goal is not normalization. The goal is sustained alignment between disease activity, treatment burden, and patient priorities.
Monitoring is not just laboratory surveillance
Laboratory data are necessary. They are not sufficient.
Longitudinal monitoring integrates:
- hemoglobin trend rather than isolated values
- hemolysis markers interpreted in context
- frequency and severity of exacerbations
- transfusion exposure over time
- cold-induced circulatory symptoms
- functional limitation
- treatment toxicity and cumulative risk
- thrombotic risk indicators and interval vascular events
Quality-of-life studies confirm that patient-reported burden often diverges from laboratory indices.5
Laboratory values describe hemolysis. They do not describe life.
Stability therefore means predictable, acceptable disease impact within the patient’s lived reality, not unchanged laboratory numbers.
Monitoring cadence should match disease behavior
There is no universal follow-up interval in CAD.6
Cadence should reflect:
- disease activity
- recent therapy changes
- prior exacerbations
- comorbid risk
- reliability of follow-up
Closer monitoring is appropriate during:
- treatment initiation or withdrawal
- recent exacerbation
- intercurrent illness
- perioperative periods
Less frequent follow-up may be appropriate during sustained stability when patients are reliable reporters and know which symptoms should prompt reassessment.
In practice, many patients in sustained stability are followed every 3–6 months, with closer monitoring during therapy changes, exacerbations, or intercurrent illness.
Stability does not always mean “no treatment”
Patients may be:
- stable on therapy
- stable because therapy is working
- stable without therapy
These states are not interchangeable.7
Stability maintained by pharmacologic suppression carries different monitoring implications than stability reflecting intrinsic disease tempo. Failure to distinguish them can lead either to unnecessary continuation of therapy or premature discontinuation.
Anticipating relapse is part of expert care
Relapse is common and expected rather than exceptional.8
Preparation reduces crisis decision-making. Anticipatory planning includes:
- educating patients about warning signs
- defining thresholds for reassessment
- documenting prior effective interventions
- clarifying escalation strategies in advance
Early relapse signals may include:
- new fatigue or reduced stamina
- dark urine
- worsening cold-induced symptoms
- rising transfusion need
Expert care plans for relapse. It does not wait for it.
Monitoring depends on the treatment strategy
Surveillance should follow mechanism.
- Complement-directed therapy:
- monitor hemolysis control, infection risk, and vaccine status
- improved hemoglobin during complement inhibition reflects pharmacologic blockade rather than clonal remission and should not be interpreted as disease resolution
- Clone-directed therapy:
- monitor cytopenias, immune suppression, delayed response, and relapse
- Supportive strategies:
- monitor trajectory and triggers
Therapy-specific monitoring reflects therapy-specific risk.9
Monitoring should reflect what could change management, not simply what can be measured.
Avoiding therapeutic inertia and therapeutic overreach
Two opposing errors threaten long-term care.
Therapeutic inertia
gradual worsening is tolerated because it is familiar
Therapeutic overreach
expected fluctuation triggers unnecessary escalation
Both are recognized pitfalls in chronic hemolytic disorders.10
CAD is particularly vulnerable to inertia because hemoglobin may stabilize at a new, lower baseline that becomes psychologically normalized. What once would have prompted treatment may slowly come to be accepted.
Expert monitoring navigates between extremes by asking:
- Has the biology changed?
- Has patient experience changed?
- Has risk–benefit balance shifted?
Management changes should respond to meaningful clinical change, not expected fluctuation.
Reassessment is a longitudinal skill
Reassessment is not an admission of failure. It is routine longitudinal care.
Effective reassessment means:
- revisiting goals
- confirming biologic fit
- updating decisions when circumstances change
- normalizing course correction
A plan that was appropriate two years ago can become inappropriate without ever having been wrong.
The patient’s life changes, even if the disease does not
Over time, patients age, change jobs, develop comorbidities, undergo surgery, or enter new life stages.
Disease burden and treatment tolerance may shift independently of hemolysis markers.11
Long-term care succeeds when it adapts to the person, not just the pathology.
Documentation supports continuity and safety
CAD management often spans clinicians and settings.
Essential documentation includes:
- confirmed diagnosis and dominant mechanism
- prior therapies and responses
- known triggers
- monitoring parameters
- action thresholds
- patient goals
- perioperative precautions
Documentation is anticipatory planning made durable.
Explicit principle
Long-term management in cold agglutinin disease is not about maintaining control at all costs.
It is about maintaining alignment:
- between biology and therapy
- between disease activity and treatment burden
- between clinical strategy and patient priorities
Expert care monitors for meaningful change, anticipates relapse without alarm, reassesses without blame, and adjusts strategy with restraint.
Success is measured not by absence of fluctuation, but by the patient’s ability to live predictably, safely, and well alongside .a disease that rarely disappears but can often be managed thoughtfully over time.
Test your thinking
A short, judgment-focused quiz on long-term management & monitoring in cold agglutinin disease.
Evidence Box
Evidence anchors for long-term CAD management
Disease course and burden
- CAD is characterized by chronic complement-mediated hemolysis with intermittent flares and a high prevalence of persistent anemia and fatigue.12
- Hemolysis is typically ongoing year-round, but symptom burden often fluctuates with ambient temperature because cold exposure enhances IgM binding, complement activation, and cold-induced circulatory symptoms.13
- In a longitudinal cohort, 72% of patients experienced at least one severe anemia episode within the first year of follow-up, and ≥65% required transfusion during their disease course.14
- Observational series demonstrate substantial transfusion burden, often averaging more than one transfusion per patient-year and substantially higher in tertiary-center cohorts.15
Complications
- CAD is associated with a markedly increased risk of thromboembolic events, including venous, arterial, and cerebrovascular thromboses.16
- In matched-cohort analyses, thrombosis risk is approximately 1.8–1.9-fold higher than in controls.17
- Risk appears to relate more closely to hemolytic activity and LDH elevation than to hemoglobin level alone, suggesting complement-mediated endothelial activation as a key driver.18
Therapeutic response and durability
- Phase 3 trials (CARDINAL and CADENZA) showed that C1s inhibition with sutimlimab rapidly increased hemoglobin and improved hemolysis markers during the initial 26-week treatment period.19
- Long-term extension data demonstrate sustained disease control and acceptable safety for at least 2 years of continuous therapy.20
- Patient-reported outcomes, including fatigue and global health measures, remain substantially improved during ongoing treatment.21
- After treatment discontinuation, quality-of-life scores tend to drift toward baseline, indicating that benefit depends on continued complement inhibition.22
- Smaller long-term cohorts extending beyond 2 years show similar safety and efficacy signals.23
Therapeutic limitations
Despite encouraging extension data, long-term safety conclusions remain constrained by small sample sizes, limited follow-up, and the rarity of CAD.
Clinical Practice Guideline Box
Guidelines and Consensus Principles for Longitudinal CAD Care
Recommendations below synthesize formal guideline statements and international consensus on autoimmune hemolytic anemia, supplemented by CAD-focused reviews, because high-grade trial evidence remains limited in this rare disease.
1. Monitor disease trajectory, not single values
CAD is chronic and fluctuating, so monitoring should emphasize trends in hemoglobin, hemolysis markers, and clinical status rather than isolated laboratory results.
Hemolysis parameters (LDH, bilirubin, haptoglobin, reticulocytes) should be interpreted alongside symptoms, functional impact, and comorbidities when deciding whether management should change.
Supported by international AIHA consensus recommendations and BSH AIHA guideline statements on chronic disease monitoring.
2. Anticipate complications
Patients with CAD have increased thromboembolic risk.
Risk relates to active hemolysis and complement activation and may exceed that predicted by anemia severity alone.
Thromboprophylaxis is recommended for hospitalized patients with acute hemolysis and should be considered in high-risk clinical settings such as surgery or severe exacerbations.
Supported by BSH AIHA guideline and international consensus recommendations.
3. Match monitoring intensity to therapy and disease activity
Follow-up cadence should reflect hemolytic activity, recent exacerbations, comorbid risk, and therapy phase, with closer monitoring during treatment initiation, withdrawal, intercurrent illness, or perioperative periods.
Patients receiving complement inhibitors require surveillance for hemolysis control, infection risk, and vaccine protection.
Patients receiving clone-directed therapy require monitoring for cytopenias, immunosuppression, delayed response, and relapse.
Supported by international consensus guidance and BSH recommendations on treatment-phase–dependent monitoring.
4. Treat the mechanism driving disease
Primary CAD is a clonal IgM-mediated, complement-driven hemolytic disorder. Disease-directed therapy should target either the pathogenic clone or complement pathway rather than rely on nonspecific immunosuppression alone.
Supportive care and corticosteroids alone have limited durable benefit in CAD.
Disease-modifying therapy is generally reserved for clinically significant anemia, transfusion dependence, or disabling cold-induced circulatory symptoms.
Supported by BSH guideline recommendations and international consensus statements on CAD pathophysiology and treatment.
5. Recognize chronicity and uncertainty in longitudinal care
Most adults with CAD have an underlying clonal lymphoproliferative disorder and follow a chronic, relapsing course. Expectations should therefore be framed around long-term disease control, not cure.
Validated biomarkers predicting response durability or relapse remain limited, and access to newer therapies varies across settings, making individualized risk–benefit assessment essential.
Supported by international consensus guidance and contemporary CAD reviews.
