Note: The video and audio linked above were generated with the assistance of AI. Clinical accuracy has been reviewed, but no AI-generated content can be guaranteed to be fully error-free.
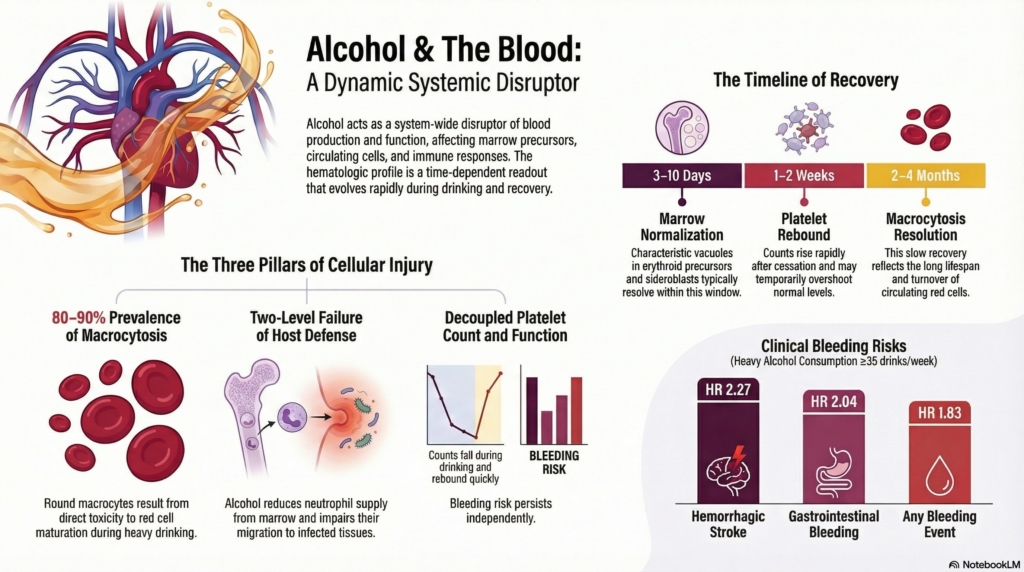
Alcohol is best understood not as a cause of a single hematologic disorder, but as a system-wide disruptor of hematopoiesis and hemostasis. It acts simultaneously at the level of marrow precursors, circulating blood cells, plasma proteins, and immune function. What makes it distinctive is not just the breadth of injury, but its tempo: the hematologic picture evolves over days to weeks with intoxication and abstinence. A complete blood count in this setting is therefore not a static diagnostic snapshot, but a time-dependent readout of recent exposure and recovery.
Bone marrow and hematopoiesis
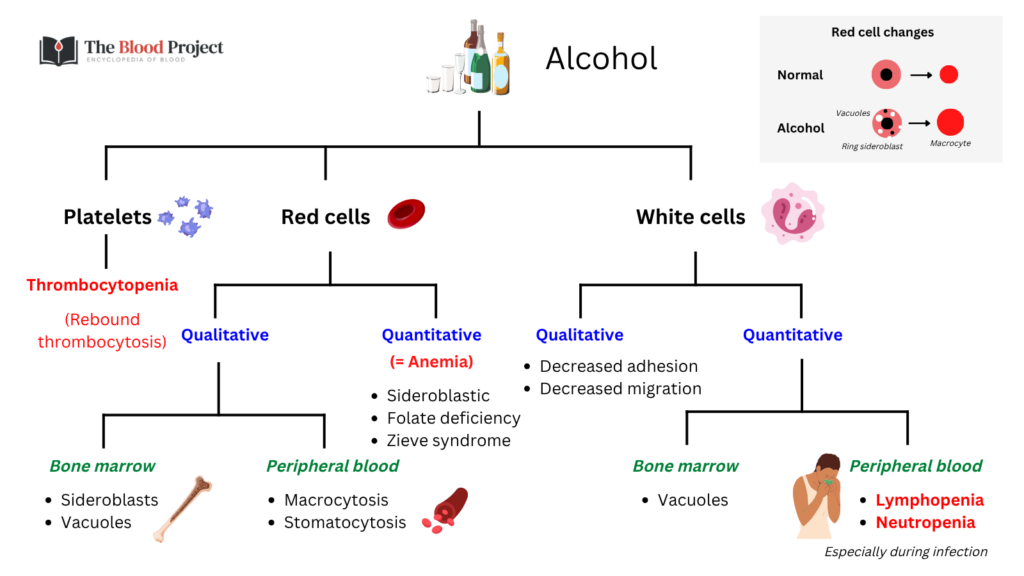
Alcohol directly suppresses hematopoiesis and produces characteristic morphologic abnormalities in marrow precursors. Severe exposure reduces marrow cellularity and may result in anemia, neutropenia, thrombocytopenia, or pancytopenia, yet these abnormalities are often reversible with abstinence, reflecting injury at the level of differentiating precursors rather than irreversible stem cell failure.
A defining feature is vacuolization of early erythroid precursors, particularly pronormoblasts. These vacuoles typically appear 5–7 days after heavy drinking and resolve within 3–10 days of abstinence, occasionally persisting up to two weeks. Their formation reflects direct toxic effects of ethanol on developing cells rather than nutritional deficiency, and their disappearance after cessation reinforces the reversibility of the process.
Alcohol also produces sideroblastic change and, in some patients, megaloblastic maturation. Importantly, marrow cultures in alcohol-exposed individuals may remain normal, suggesting that the toxic effect is exerted on more mature precursor compartments, which explains why hematopoiesis can recover rapidly once exposure stops.
Marrow findings in alcohol use often reflect the last several days of exposure rather than an underlying disorder. Apparent abnormalities may resolve with time and abstinence.
Red blood cells
Red cell abnormalities are among the most common hematologic manifestations of alcohol use and arise from overlapping mechanisms: direct toxicity, nutritional deficiency, impaired heme synthesis, and altered membrane structure.
The most recognizable pattern is macrocytosis, which frequently occurs without anemia and often reflects a direct toxic effect of alcohol on erythroid maturation rather than folate deficiency or liver disease.
- 80–90% of individuals consuming >80 g ethanol daily have macrocytosis
- alcohol is the most common cause of macrocytosis in several clinical series
These macrocytes are typically round and uniform, in contrast to the oval macrocytes of megaloblastic anemia. One reflects direct toxicity, the other disordered DNA synthesis. Macrocytosis resolves slowly, over 2–4 months of abstinence, consistent with the lifespan and turnover of circulating red cells, and therefore serves as a marker of sustained exposure.
Megaloblastic anemia is also common, particularly in hospitalized or poorly nourished alcoholics. In some series, it accounts for approximately 40% of anemia cases among alcoholics, reflecting folate deficiency. Although folate deficiency is less common today in the general population because of food fortification, it remains a relevant cause in alcohol use, particularly in those with poor nutritional intake. Alcohol contributes not only by depleting folate stores, but also by impairing the marrow’s ability to utilize folate effectively, even when it is present. This distinction explains why folate replacement alone may not fully correct the anemia without abstinence.
Alcohol also disrupts heme synthesis, producing ring sideroblasts. These are typically transient morphologic changes rather than a true sideroblastic anemia, reflecting a reversible disturbance in erythroid metabolism. They often resolve within 5–10 days of abstinence, before a sustained anemia phenotype has time to develop.
Less commonly, alcohol produces hemolysis through membrane and metabolic effects. This includes stomatocyte-associated hemolysis, spur-cell anemia in advanced liver disease, and metabolic hemolysis during withdrawal. In some patients, a syndrome of hemolysis, hyperlipidemia, and liver dysfunction has been described (often termed Zieve syndrome), although its mechanistic unity remains debated in the literature.
The broader lesson is that anemia in alcohol use is rarely singular. It is typically multifactorial, combining impaired production, nutritional deficiency, blood loss, and, in some cases, increased destruction.
White blood cells and host defense
Alcohol impairs host defense at multiple levels, affecting both the production and function of leukocytes, particularly neutrophils. The consequence is not simply a low white count, but a failure of the neutrophil system.
Alcohol suppresses granulopoiesis, reduces marrow neutrophil reserves, and disrupts the ability to mount an emergency response to infection. At the same time, it impairs neutrophil adhesion and migration, limiting delivery of cells to infected tissues. The result is a two-level failure: reduced supply and impaired deployment.
Clinically, this may present as transient neutropenia during infection. In one series, half of alcoholic patients with severe bacterial infection were neutropenic at presentation, and the remainder developed neutropenia within 24–48 hours, followed later by rebound leukocytosis over the next several days.
The underlying biology is more complex. Alcohol disrupts both baseline granulopoiesis and the signaling pathways required for stress-induced neutrophil production, leading to impaired responses precisely when demand is highest.
Alcohol also impairs macrophage function, reducing bacterial clearance in tissues such as lung and peritoneum. These defects are often reversible, with improvement within about a week of abstinence.
Alcohol also affects lymphocytes, although less prominently than neutrophils. Lymphopenia may occur in the setting of severe alcohol use, but the more important effect is functional. Alcohol impairs lymphocyte responses to new antigens and disrupts T-cell–mediated immunity, limiting the ability to mount an effective adaptive immune response.
Together with neutrophil dysfunction, these changes impair host defense at multiple levels, affecting both the immediate and sustained response to infection. A normal or even elevated white count does not necessarily imply effective immunity.
Platelets
Platelet abnormalities in alcohol use involve both decreased production and impaired function. Alcohol suppresses megakaryopoiesis, shortens platelet survival, and interferes with platelet activation pathways.
This produces a characteristic temporal pattern: platelet counts fall during active drinking and then rise rapidly after cessation, sometimes reaching markedly elevated levels during recovery, in keeping with the short lifespan of circulating platelets.
This produces a characteristic temporal pattern: platelet counts fall during active drinking and then rise rapidly after cessation, sometimes reaching markedly elevated levels during recovery, in keeping with the short lifespan of circulating platelets. Counts decline during exposure, recover quickly with abstinence, and may overshoot before stabilizing.
In addition to quantitative changes, ethanol directly inhibits platelet responses to physiologic agonists, contributing to bleeding risk independent of platelet count.
Platelet count and function do not track together. Alcohol impairs both, so clinical risk cannot be inferred from the count alone.
Coagulation, fibrinolysis, and bleeding
Alcohol alters multiple components of hemostasis, including coagulation factors, platelet function, and fibrinolysis. The net effect depends on dose and context, with heavy alcohol use associated with increased bleeding risk.
Epidemiologic data illustrate this clearly. In a cohort of 10,259 individuals, consumption of ≥35 drinks per week was associated with increased risk of:
- hemorrhagic stroke (HR 2.27)
- gastrointestinal bleeding (HR 2.04)
- any bleeding (HR 1.83)
Mechanistically, alcohol influences fibrinolysis in a bidirectional manner. Moderate intake may enhance fibrinolytic activity, whereas heavy intake and its metabolites can suppress it, reflecting changes in regulatory proteins such as tissue plasminogen activator and its inhibitors. At the same time, alcohol impairs platelet function and alters hepatic synthesis of clotting proteins.
The result is not a simple anticoagulated state, but a destabilized hemostatic system, in which the balance between thrombosis and bleeding becomes unpredictable and dose-dependent.
Iron metabolism and diagnostic pitfalls
Alcohol affects iron balance in opposing ways. Chronic use may lead to iron deficiency through gastrointestinal bleeding, but it may also promote iron overload through increased absorption and ineffective erythropoiesis.
This creates diagnostic challenges:
- serum iron may fluctuate with drinking and abstinence
- ferritin may be elevated due to liver disease rather than iron stores
As a result, standard iron studies may be difficult to interpret in alcohol users, and iron abnormalities must be interpreted in clinical context.
Dynamic clinical patterns
The most important unifying concept is temporal instability. Hematologic abnormalities in alcohol use evolve predictably with exposure and abstinence.
- marrow vacuoles and sideroblasts resolve over days to weeks
- neutropenia during infection may transition to leukocytosis over 5–10 days
- platelet counts fall during intoxication and rebound during recovery
- macrocytosis persists for months, reflecting cumulative exposure
This means that the timing of observation is often more informative than the abnormality itself. A CBC drawn during intoxication and one drawn a week later may tell entirely different stories.
Table. Alcohol and the hematologic system: dynamic, reversible, and time-dependent changes across cell lines
🧬 Bone marrow
| Count | Form / fxn | Why | Time | Clinical |
|---|---|---|---|---|
| • ↓ cellularity (some) • ± pancytopenia | • vacuolated precursors • ring sideroblasts • megaloblastoid change | • direct EtOH toxicity • mito injury (iron handling) • ↓ DNA synthesis | • vacuoles: 3–7 d • sideroblasts: 5–10 d | • mimics MDS • reflects recent exposure • reversible |
🩸 Red cells
| Count | Form / fxn | Why | Time | Clinical |
|---|---|---|---|---|
| • Hb ↓ or nl • MCV ↑ (<110) | • round (toxic) macrocytes • anisocytosis • poikilocytosis • ± hemolysis (stomatocytes, spur, Zieve) | • membrane toxicity • lipid alteration • ± folate deficiency • impaired folate use | • resolves 2–4 mo • reflects RBC lifespan | • often only clue • anemia often multifactorial • distinguish from megaloblastic |
🧪 White cells
| Count | Form / fxn | Why | Time | Clinical |
|---|---|---|---|---|
| • neutropenia • ↓ reserve • rebound leukocytosis | • maturation arrest (myelocyte) • ↓ adhesion (Mac-1) • ↓ delivery to infection | • marrow suppression • cytokine disruption • trafficking defect | • neutropenia during infection • rebound 5–10 d | • infection risk • counts ≠ function • impaired innate defense |
🧩 Lymphocytes
| Count | Form / fxn | Why | Time | Clinical |
|---|---|---|---|---|
| • mild ↓ (variable) | • ↓ T-cell function • impaired antigen response | • immune dysregulation • adaptive defect | • variable • improves with abstinence | • impaired adaptive immunity • infection susceptibility |
🧫 Platelets & coagulation
| Count | Form / fxn | Why | Time | Clinical |
|---|---|---|---|---|
| • ↓ platelets (active) • rebound ↑ | • ↓ aggregation • impaired function | • ↓ megakaryocytes • ↓ survival • ethanol inhibition | • nadir during drinking • rebound 3–14 d | • count ≠ bleeding risk • dysfunction persists |
| • ± factor changes | • impaired hemostasis • altered fibrinolysis | • platelet dysfunction • liver dysfunction | • variable | • bleeding risk multifactorial |
Closing perspective
Alcohol produces a constellation of hematologic effects that are broad, overlapping, and often reversible. The most common clue is macrocytosis. The most clinically important consequences are infection and bleeding. But the most transferable insight is this: in alcohol-related hematology, the timing of the observation is often more diagnostically meaningful than the finding itself, because many abnormalities move toward normal with abstinence, and those that do not are the ones that demand alternative explanation.
Further reading
- Shi et al. Alcohol Abuse and Disorder of Granulopoiesis. Pharmacol Ther. 2019;198: 206–219.
- Larkin and Watson-Williams. Alcohol and the Blood. Med Clin North Am. 1984;68(1):105-20.
- Chanarin J. Alcohol and the blood. Br J Haem. 1979;42:333-336.
- Lindenbaum J. Hematologic Complications of Alcohol Abuse. Semin Liver Dis. 1987;7(3):169-81.
- Ballard HS. The Hematological Complications of Alcoholism. Alcohol Health Res World. 1997;21(1):42-52.
