How laboratory patterns, temperature, complement, and clonality converge
Note: The video and audio linked above were generated with the assistance of AI. Clinical accuracy has been reviewed, but no AI-generated content can be guaranteed to be fully error-free.
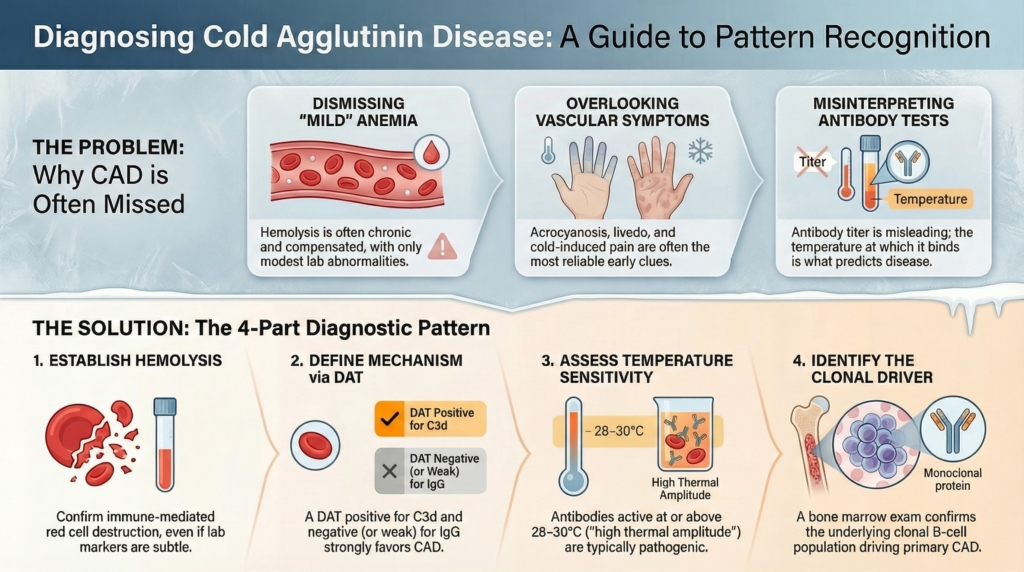
Diagnosis in cold agglutinin disease (CAD) is not established by a single test. It emerges from a pattern. Hemolysis may be incomplete, antibody titers misleading, and symptoms disproportionate to laboratory abnormalities. Accurate diagnosis requires integrating evidence of immune-mediated hemolysis, complement involvement, temperature dependence, and an underlying clonal B-cell process, in that order.1
This sequence matters: hemolysis establishes relevance, complement defines mechanism, temperature explains behavior, and clonality determines chronicity and classification.
Treat this as a hierarchy, not a checklist: start by proving hemolysis is clinically relevant, then confirm complement-mediated mechanism, then explain temperature dependence, and only then pursue clonality to establish chronicity and classification.
For example, a patient with fatigue, mild anemia, and cold-induced finger discoloration might first be confirmed to have clinically relevant hemolysis, then shown to have complement-positive DAT, then demonstrated to have temperature-dependent antibody activity, and only afterward evaluated for a clonal B-cell population. The diagnosis emerges as each layer aligns.
Recognizing hemolysis — often incomplete, often underestimated
In CAD, vascular symptoms are not ancillary. Acrocyanosis, livedo, Raynaud-like pain, and cold-induced dysesthesia often reflect complement-mediated agglutination and vascular dysfunction rather than anemia itself. In some patients, these features provide the earliest and most reliable diagnostic clues, particularly when laboratory markers of hemolysis appear modest.2
Hemolysis in CAD is frequently chronic, compensated, and laboratory-subtle, leading to underrecognition when clinicians expect dramatic biochemical abnormalities.3
Symptoms often reflect complement activation and microvascular dysfunction more than anemia severity.4 For that reason, the hemolysis pattern in CAD is often best recognized by combining symptoms and signs with laboratory evidence, rather than expecting a dramatic biochemical signature in isolation.5
Typical laboratory features include:6
- anemia that is often mild to moderate rather than profound
- LDH and indirect bilirubin that may be only modestly elevated
- haptoglobin that may be low or borderline rather than absent
- reticulocyte responses that are variable and sometimes blunted
Fatigue and functional impairment commonly exceed what hemoglobin values alone would predict.7
The direct antiglobulin test (DAT): the diagnostic pivot
In CAD, the DAT pattern is more informative than DAT positivity alone. When CAD is suspected, monospecific DAT testing should be requested because polyspecific DAT alone cannot distinguish complement-mediated hemolysis from IgG-mediated disease.
Complement deposition, rather than IgG binding, defines the disease mechanism and distinguishes CAD from warm autoimmune hemolytic anemia.8
In CAD, complement is typically detected as C3d because C3b deposited on the red cell surface is progressively cleaved during circulation, leaving C3d as the stable remnant on surviving cells. This helps explain how DAT findings can remain strongly complement-positive even when hemolysis is chronic and partially compensated.9
Typical features include:10
- DAT positivity for C3d
- IgG that is usually negative or only weakly positive
- isolated C3 positivity that strongly favors CAD
Mixed DAT patterns require careful interpretation. True IgG plus C3 positivity raises the possibility of mixed autoimmune hemolytic anemia, which carries different diagnostic and therapeutic implications.11
Complement studies can provide supportive evidence of ongoing classical pathway activation. In some patients, complement components are depleted, including very low C4 (and sometimes low C3), consistent with active complement consumption in CAD biology.12
Disproportionately low C4 with relatively preserved C3 reflects classical pathway activation, the complement pathway that characterizes CAD, and helps distinguish it from alternative pathway–driven disorders.
Cold agglutinins: presence is not disease
Cold agglutinins are common in the general population, particularly at low titers. CAD should not be diagnosed based on their presence alone. Clinical relevance depends on antibody behavior and downstream complement effects.13
Low-titer cold agglutinins are frequently incidental, and antibody titer alone does not define pathogenicity. Although titers above traditional thresholds (for example, >1:64 at 4°C) are often reported as “significant,” this remains an imperfect surrogate for clinical relevance because temperature dependence and complement engagement matter more than quantity.14
Clinically significant CAD usually involves monoclonal IgM, with antibody activity at physiologic temperatures playing a dominant role.15
Thermal amplitude: the most underused diagnostic concept
Thermal amplitude, the highest temperature at which the antibody binds red cells, is the strongest predictor of clinical significance. It explains seasonal variability, persistence of symptoms after cold exposure, and poor correlation between antibody titer and disease severity.16
Antibodies active near core body temperature are typically pathogenic and correlate with greater hemolysis and symptom burden.17 The reason is anatomical: higher thermal amplitude means IgM binding persists at temperatures encountered beyond the periphery, expanding the zone where complement can be activated and making hemolysis less dependent on extreme cold exposure alone.18
A low-titer antibody active at higher temperatures may cause more severe disease than a high-titer antibody binding only at low temperatures, explaining why antibody quantity alone is misleading.19
When thermal amplitude testing is unavailable, clinical behavior serves as a surrogate, including persistence beyond winter months or symptoms triggered by mild cold exposure.20
Distinguishing primary CAD from secondary cold agglutinin syndrome
This distinction alters diagnostic evaluation, prognosis, and management. Primary CAD is a chronic clonal disorder; secondary cold agglutinin syndromes are reactive and often transient.21
Primary CAD is characterized by:22
- chronic course
- clonal bone marrow B-cell disorder
- persistent complement-mediated hemolysis
Secondary cold agglutinin syndromes are associated with infection, autoimmune disease, or overt lymphoma and often resolve with treatment of the underlying condition.23
Infection may also unmask previously subclinical primary CAD by transiently increasing complement availability, so longitudinal follow-up is often required to distinguish persistent clonal disease from self-limited post-infectious hemolysis.
When available, antibody specificity can also support classification. Infection-associated cold agglutinin syndromes are often polyclonal and may show anti-I specificity with Mycoplasma pneumoniae, whereas EBV-associated cases may show anti-i specificity, reinforcing that “post-infectious” disease is biologically heterogeneous.24
Bone marrow evaluation: diagnosing the clone
Bone marrow examination in cold agglutinin disease is performed to identify the antibody-producing B-cell clone, not to stage lymphoma. The diagnostic goal is to uncover the biological driver of hemolysis rather than to define tumor burden or nodal disease.25
Clonal B-cell populations are present in most patients with primary CAD. These clones are typically low-grade, indolent, and confined to the bone marrow rather than presenting as overt lymphadenopathy.26
Marrow involvement is often subtle and may be missed without careful histologic, immunophenotypic, and molecular evaluation. Flow cytometry is particularly useful because light-chain restriction and characteristic surface marker patterns can identify small clonal B-cell populations that are not evident morphologically. In many cases, the lymphoproliferative disorder does not meet criteria for a defined lymphoma subtype at presentation.27
When present, the clonal population most often shows lymphoplasmacytic or marginal zone–like features, supporting the concept that primary CAD is driven by a distinct marrow-based lymphoproliferative process rather than classic nodal lymphoma.28
Overt lymphoma is uncommon at the time of CAD diagnosis. The absence of radiographic lymphadenopathy or bulky disease should not be interpreted as evidence against primary CAD.29
Importantly, clone size correlates poorly with anemia severity. Clinical phenotype is driven more by antibody characteristics and complement engagement than by the extent of marrow infiltration. This disconnect explains why patients with minimal marrow disease may experience severe hemolysis, while others with larger clones remain relatively compensated.30
Bone marrow evaluation is recommended once CAD is suspected, but it does not always need to precede initial management. In patients with severe or rapidly progressive hemolysis, treatment may begin before full marrow characterization. In more stable patients, marrow evaluation helps clarify disease classification, informs prognosis, and guides longer-term management decisions.31
Diagnostic convergence in primary CAD
Diagnosis is established when the following signals align:
• clinically relevant hemolysis
• DAT positive for C3d (± weak IgG)
• pathogenic cold antibody with significant thermal amplitude
• monoclonal IgM and marrow-based clonal B-cell population
No single finding is sufficient. Diagnosis emerges from convergence.
Even when these signals align, pre-analytical interference can obscure or distort laboratory interpretation.
Sample handling and laboratory interference: an invisible diagnostic pitfall
Cold agglutinins can interfere with routine blood counts if samples are allowed to cool before analysis. In some cases, visible clumping in the collection tube that resolves after warming is itself the first diagnostic clue. This pre-analytical issue can distort results and delay diagnosis.32
- red cell agglutination may falsely lower RBC count
- MCV may appear spuriously elevated
- MCHC may be artifactually increased
- automated analyzers may flag clumping or report implausible indices
When CBC parameters appear internally inconsistent, warming the sample and repeating analysis is essential. Failure to recognize cold agglutinin interference can obscure hemolysis and mislead interpretation.33
Common diagnostic pitfalls
Diagnostic delay in cold agglutinin disease is usually conceptual rather than technical. Errors most often arise from misinterpreting patterns, underweighting symptoms, or applying inappropriate diagnostic frameworks.34
Common pitfalls include:35
- dismissing symptoms because anemia appears mild:
In CAD, fatigue, cold intolerance, pain, and functional limitation may be driven by complement activation and microvascular dysfunction rather than hemoglobin level alone. Symptom burden often exceeds what would be predicted by laboratory measures of anemia. - relying on cold agglutinin titer alone:
Cold agglutinin titers are an unreliable indicator of pathogenicity. Antibody behavior at physiologic temperatures and complement engagement are far more important determinants of clinical relevance than titer magnitude. - misclassifying CAD as warm autoimmune hemolytic anemia due to weak IgG DAT positivity:
Weak or borderline IgG positivity on DAT does not exclude CAD and may reflect nonspecific binding or technical factors. Interpretation should prioritize complement deposition and the overall clinical pattern. True IgG plus C3 positivity raises the possibility of mixed autoimmune hemolytic anemia, which requires different diagnostic and therapeutic reasoning. - assuming absence of overt lymphoma excludes CAD:
Most patients with primary CAD harbor a low-grade, marrow-based clonal B-cell disorder without bulky lymphadenopathy or radiographic evidence of lymphoma. Normal imaging does not rule out clonal disease. - failing to link cold exposure, seasonality, or environmental triggers to symptoms:
Disease activity that worsens with cold exposure, winter months, or air-conditioned environments provides an important diagnostic clue to temperature-dependent antibody activity and higher thermal amplitude.
Diagnosis as pattern recognition, not confirmation
CAD is diagnosed when multiple signals align: immune-mediated hemolysis, complement deposition on DAT, temperature-dependent IgM activity, and an underlying clonal B-cell disorder. No single test is sufficient. Sometimes the diagnosis converges in a single encounter, but often it requires longitudinal reassessment as persistence, temperature dependence, and clonality become clearer over time.36
Test your thinking
A short, judgment-focused quiz on the diagnosis of cold agglutinin disease.
