What CAD is—and what it is not
Note: The video and audio linked above were generated with the assistance of AI. Clinical accuracy has been reviewed, but no AI-generated content can be guaranteed to be fully error-free.
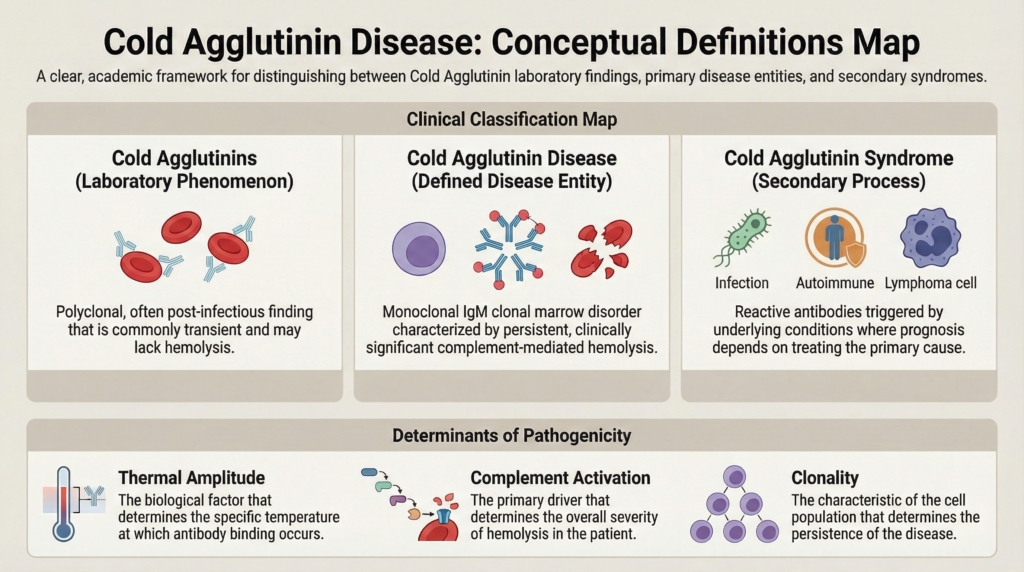
What is cold agglutinin disease?
Primary CAD is a clonal cold-antibody autoimmune hemolytic anemia in which a pathogenic, usually monoclonal IgM autoantibody binds red blood cells at lower temperatures and activates the classical complement pathway, leading predominantly to C3-mediated extravascular hemolysis.1
Conceptual anchor:
The antibody initiates the process; complement largely determines the hemolytic phenotype.
Key defining elements
- presence of a monoclonal IgM autoantibody, usually κ-restricted2
- classical complement activation as the dominant effector mechanism3
- C3 deposition on red blood cells4
- predominantly hepatic extravascular clearance of complement-opsonized red blood cells5
- a chronic or relapsing disease course in most primary cases6
CAD is not simply “autoimmune hemolytic anemia triggered by cold.” It is a mechanistically distinct entity with its own biology, kinetics, and therapeutic vulnerabilities.7
What is a cold agglutinin?
A cold agglutinin is an antibody that binds red blood cell antigens more avidly at temperatures below core body temperature.8
Cold agglutinins vary widely in clinical significance. Some are incidental laboratory findings, while others are pathogenic because they bind red cells at physiologically relevant temperatures and activate complement.
In clinically significant cold-antibody hemolysis, the antibody is usually IgM, binds red cells in cooler peripheral circulation, may dissociate on rewarming, and can leave behind complement fragments that continue to mark the red cell for destruction.
Cold agglutinins versus cold agglutinin disorders
Cold agglutinins are commonly detectable in human sera, but most are low-titer, polyclonal, and clinically insignificant.9 The presence of a cold agglutinin does not equal cold agglutinin disease, and it does not necessarily imply clinically significant hemolysis.10
Important distinctions:
Incidental or transient cold agglutinins11
- often polyclonal
- commonly post-infectious
- usually self-limited
- may cause mild or no hemolysis
- do not necessarily define a clinical syndrome
Secondary cold agglutinin syndrome
- cold agglutinin-mediated hemolysis occurring secondary to another condition
- may follow infection or occur with autoimmune disease or overt lymphoid malignancy
- may be transient or persistent depending on the underlying cause
- management focuses on the associated condition and supportive care, with CAD-directed therapy considered only when clinically appropriate
Primary cold agglutinin disease12
- typically monoclonal
- persistent
- complement-driven hemolysis
- usually associated with a clonal bone marrow disorder
Why clonality matters:13
Clonality implies a persistent antibody source. This is why primary CAD is usually chronic, while many incidental or infection-associated cold agglutinins are self-limited.
Failure to separate incidental or transient cold agglutinins, secondary cold agglutinin syndromes, and primary clonal CAD has historically led to diagnostic confusion and inappropriate therapy.
Table. Distinguishing cold agglutinins, secondary cold agglutinin syndrome, and primary CAD
| Category | Antibody pattern | Typical context | Hemolysis | Expected trajectory | Clinical meaning |
|---|---|---|---|---|---|
| Incidental or transient cold agglutinins | often polyclonal, low titer | healthy individuals or after infection | none or mild | usually self-limited | a laboratory finding, not necessarily disease |
| Secondary cold agglutinin syndrome | often polyclonal or context-dependent | infection, autoimmune disease, overt lymphoid malignancy | variable, sometimes clinically significant | depends on the underlying condition | cold-antibody hemolysis occurring in another disease context |
| Primary CAD | usually monoclonal IgM, often κ-restricted | clonal marrow B-cell process | chronic complement-mediated hemolysis | persistent or relapsing | a distinct clinicopathologic entity |
| Mixed AIHA phenotype | IgG and C3 often both present on DAT | overlapping warm and cold mechanisms | variable | depends on dominant mechanism(s) | boundary condition requiring caution with classic CAD assumptions |
Primary CAD versus secondary cold agglutinin syndrome
Primary CAD14
- chronic clonal disorder
- IgM produced by an indolent B-cell clone
- complement-mediated hemolysis intrinsic to the disease
- no overt associated condition driving the cold antibody process
Secondary cold agglutinin syndromes
Occurs in association with:15
- acute infections
- autoimmune diseases
- overt lymphoid malignancies
In secondary cold agglutinin syndrome, the cold antibody process is part of another disease context. In primary CAD, the cold antibody is produced by the clonal marrow process that defines the disease.
In secondary syndromes, treatment primarily targets the underlying condition. In primary CAD, therapy is directed toward complement activation, the clonal source, or both.
This distinction materially affects prognosis, evaluation for associated disease, monitoring strategy, and treatment selection.16
Thermal amplitude
Thermal amplitude is the highest temperature at which a cold agglutinin binds red blood cells.17
Why it matters18
- higher thermal amplitude → greater physiologic relevance
- is often more informative than titer alone in judging clinical relevance
- explains severe disease despite modest antibody levels
Antibodies capable of binding at near-core temperatures interact with erythrocytes in central circulation, not just acral regions, which explains disproportionate disease severity. An antibody that binds at 30 °C may be clinically dangerous, while one that binds only at 4 °C may be biologically irrelevant.
Complement as the central effector
In CAD:19
- the antibody initiates the process
- complement determines the phenotype
Key principles20
- hemolysis is predominantly driven by classical pathway activation
- C3 deposition is dominant
- most destruction is extravascular
- terminal complement lysis is usually limited
This creates an important temporal uncoupling: the IgM antibody may detach as red cells return to warmer central circulation, but complement fragments can remain on the red-cell surface and continue to mark the cell for destruction.
Limited terminal pathway activation helps explain why CAD typically presents with chronic anemia rather than fulminant intravascular hemolysis. Host complement regulators on the red-cell surface restrict progression to full membrane-attack–complex formation.
This mechanism also explains therapeutic logic: proximal complement blockade can suppress hemolysis without eliminating the pathogenic clone.21
Clonality and bone marrow involvement
Most patients with primary CAD have:22
- a detectable monoclonal IgM
- an underlying indolent B-cell lymphoproliferative disorder
- disease largely confined to bone marrow
Primary CAD is therefore best understood as a clonal marrow B-cell disorder with immune-mediated hemolytic manifestations, rather than a purely reactive autoimmune condition.23
The precise classification of the underlying clone has evolved historically, underscoring that disease definitions reflect current biologic understanding rather than fixed categories.
Common clinical misconceptions
CAD is not:24
- primarily IgG mediated
- predominantly splenic in clearance
- reliably steroid responsive
- defined by antibody titer alone
- a uniform or predictable disease
Steroid responsiveness is the exception, not the rule.
Misunderstanding what CAD is not has historically driven ineffective therapy.
Core conceptual summary
Cold agglutinin disease rests on three biologic pillars:
- clonality determines persistence
- complement determines phenotype
- thermal amplitude determines pathogenicity
CAD is therefore defined as much by complement biology as by anemia itself.
Its mechanism is defined.
Its expression is variable.
Its classification is useful but not absolute.
Effective care depends on knowing which features are definitional, which are variable, and which only appear important.
Test your thinking
Test your ability to distinguish definitions, mechanisms, and diagnostic criteria in cold agglutinin disease.
Guideline perspective
How consensus documents define and diagnose cold agglutinin disease
Diagnostic definition (international consensus)
Primary CAD is defined by chronic hemolysis, a cold agglutinin titer typically ≥64 at 4 °C, characteristic direct antiglobulin test findings, and absence of an underlying associated disorder.25
DAT profile
The typical serologic pattern is a monospecific DAT strongly positive for C3d, with IgG usually negative but sometimes weakly positive.26
Titer interpretation
Titer should be reported as the reciprocal of the highest dilution producing agglutination, recognizing that rare cases of clinically significant CAD may occur with titers below 64.27
Thermal amplitude testing
Measurement of thermal amplitude is not required for diagnosis in most patients but may be useful in selected cases to distinguish pathologic antibodies from benign low-titer cold agglutinins.28
Primary vs secondary disease
The same laboratory criteria apply to secondary cold agglutinin syndrome, and clinical, histologic, and radiologic evaluation should be performed to exclude underlying malignancy or other causes.29
Underlying clonal disorder
Serum monoclonal IgMκ is detectable in most patients, and clonal B-cells are usually demonstrable in marrow using sensitive methods.30
