Two parallel processes: hemolysis and agglutination
Note: The video and audio linked above were generated with the assistance of AI. Clinical accuracy has been reviewed, but no AI-generated content can be guaranteed to be fully error-free.
Framing the clinical problem
Cold agglutinin disease is a distinct clinicopathologic entity with a heterogeneous clinical presentation. CAD is best understood as two parallel processes, complement-mediated hemolysis and cold-induced red-cell agglutination, each contributing independently to the clinical picture. Its manifestations reflect the interaction between cold-antibody activity, complement activation, and peripheral temperature gradients, resulting in symptoms that fluctuate with environment and may appear discordant with laboratory findings.1
Patients may live with symptoms for years before diagnosis is made, particularly when anemia is mild or episodic.2
Autoimmune hemolytic anemia often develops gradually over months, but in some patients onset may be abrupt and severe. Anemia severity in CAD is variable and may be mild at presentation, which contributes to delayed recognition. In children and adolescents, cold antibody hemolysis is far more often infection-associated and transient than primary CAD.
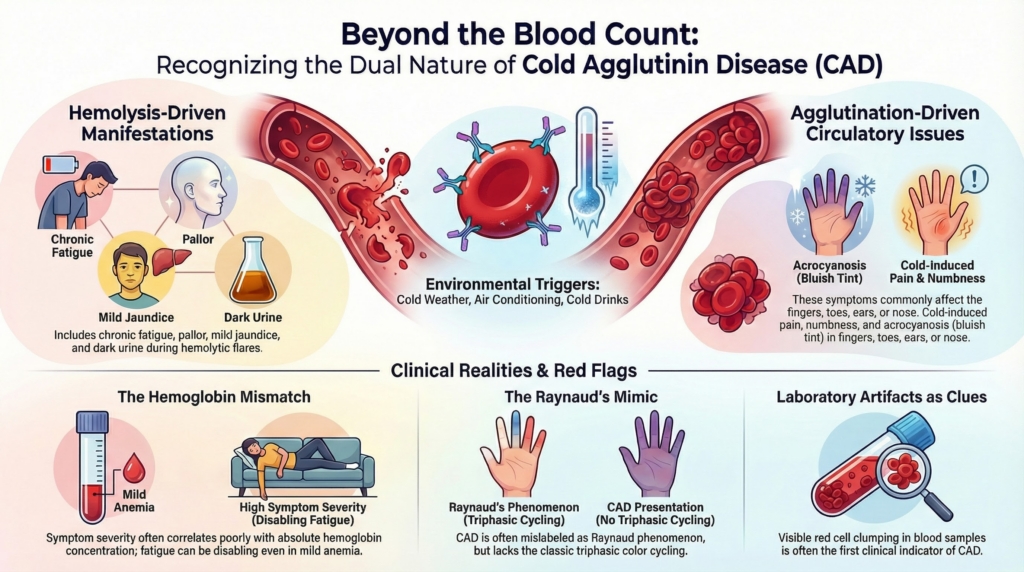
A practical way to recognize CAD early is to expect two parallel symptom streams that can be independently prominent: hemolysis-driven manifestations (anemia, jaundice, dark urine, fatigue) and agglutination-driven circulatory manifestations (acrocyanosis, cold-induced pain, acral color change).3 This dual physiology helps explain why patients with similar hemoglobin values can look dramatically different clinically. Complement-mediated hemolysis largely determines anemia severity, whereas IgM-mediated agglutination largely determines circulatory symptoms. Recognizing which stream predominates can clarify diagnostic thinking and management priorities.4
Two clinical streams in cold agglutinin disease
| Hemolysis-driven | Agglutination-driven (circulatory) |
|---|---|
| Fatigue | Acrocyanosis |
| Reduced exercise tolerance | Cold-induced pain or numbness |
| Pallor | Acral color change |
| Jaundice (often mild) | Persistent dusky discoloration |
| Dark urine (during flares) | Symptoms triggered by cold exposure |
These processes may coexist, but one often dominates the clinical picture.
A common “pre-diagnosis” vignette is a patient with mild anemia and long-standing cold-sensitive fingers labeled as Raynaud phenomenon, plus fatigue that feels out of proportion to the hemoglobin. That pattern should prompt consideration of CAD, even before the diagnosis is established.5
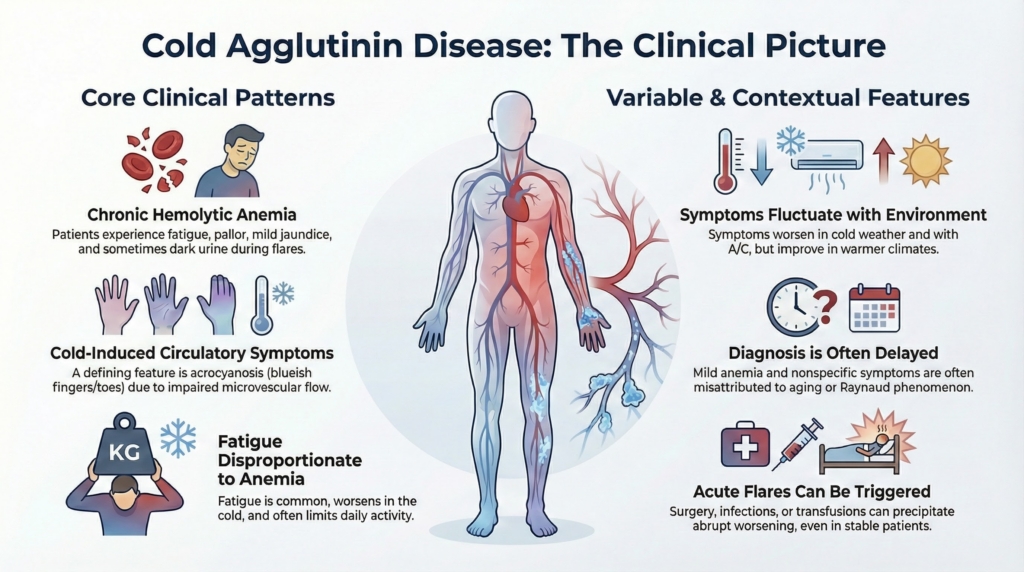
Tier 1 highlights core clinical patterns; Tier 2 highlights variability and contextual features.
Core patterns of presentation (Tier 1)
Many patients present with features of chronic hemolysis, which may be subtle or intermittent:6
- fatigue and reduced exercise tolerance
- pallor
- jaundice, often mild
- dark urine during hemolytic flares
When urine becomes frankly dark or cola-colored, this may indicate more intense hemolytic activity and should prompt assessment for acute disease activation.
Hemoglobin levels may be only modestly reduced, and symptom burden often correlates poorly with absolute hemoglobin concentration.7
A defining feature of CAD is temperature-dependent vascular symptomatology:8
- acrocyanosis of fingers, toes, ears, or nose
- Raynaud-like color changes without classic triphasic vasospasm
- numbness, pain, or stiffness in cold-exposed areas
- livedo-like or mottled cold-induced discoloration that improves with warming
In rare cases, severe agglutination-mediated circulatory impairment may be complicated by ischemic tissue injury.
These findings reflect red-cell agglutination and impaired microvascular flow rather than a primary vasospastic process. Circulatory symptoms are common and may be disabling even in moderate climates.9
A useful bedside distinction from primary Raynaud phenomenon is that CAD discoloration often remains dusky or cyanotic as long as the tissue is cold and improves with warming, rather than cycling through vasospastic phases.10 Rapid improvement with warming supports a cold-dependent agglutination mechanism.
Circulatory symptoms may dominate the clinical picture even when anemia is mild.11
Fatigue as a dominant symptom
Fatigue in CAD is common and often underestimated. It may persist despite relatively preserved hemoglobin, and it can limit daily activity.12
In patient-reported surveys, fatigue or tiredness is frequently reported before diagnosis and often carries major functional impact.13
Symptom severity often correlates poorly with hemoglobin level, suggesting that anemia alone does not capture disease impact.14
Variable and contextual features (Tier 2)
Symptoms often fluctuate:
- worse in cold weather and cold indoor environments such as air conditioning
- improved in warmer seasons
Patients commonly recognize environmental triggers before a hematologic diagnosis is established.15
Some patients experience:16
- episodic dark urine after cold exposure or febrile illness, which may accompany more intense hemolytic exacerbations
- transient worsening of anemia during infections
In post-infectious cold agglutinin syndromes, hemolysis may develop after the onset of infection and resolve as antibody levels decline.
Intercurrent infections and acute-phase responses can exacerbate hemolysis, likely in part through increased complement activation.17
Table. Clinical presentation anchors from major CAD cohorts and patient-reported studies
| Study | Population / design | Presentation findings most relevant to this essay | Best teaching use |
|---|---|---|---|
| Swiecicki et al., Blood 2013 | Single-center retrospective Mayo Clinic series of 89 patients with CAD, 1970–2012 | Median age at symptom onset 64.6 years; median age at diagnosis 71.6 years; median time from symptoms to diagnosis 37.4 months. Common symptoms during disease course included acrocyanosis 39/89 (44%), fatigue 36/89 (40%), dyspnea on exertion 19/89 (21%), and hemoglobinuria 13/89 (15%). Cold triggers were reported in 35/89 (39%). | Supports delayed recognition and the importance of both circulatory symptoms and hemolysis/fatigue in clinical presentation. |
| Berentsen et al., Blood 2020 | Multinational observational study of 232 patients with verified CAD from 24 centers in 5 countries; secondary CAS excluded by study criteria | Mean baseline hemoglobin 9.3 g/dL; median hemoglobin 9.2 g/dL; 62/232 (26.7%) had hemoglobin <8 g/dL and 89/232 (38.4%) had hemoglobin ≥10 g/dL. Among 210 patients with relevant data, cold-induced circulatory symptoms were recorded in 120/210 (51.7%) at or before diagnosis. Clinical phenotypes included hemolytic anemia with absent/grade 1 circulatory symptoms 146/210 (69.5%), hemolytic anemia with grade 2–3 circulatory symptoms 44/210 (21.0%), and circulatory symptoms with compensated hemolysis 20/210 (9.5%). | Supports the “two-stream” model: anemia/hemolysis and circulatory symptoms vary independently, and some patients have prominent circulatory symptoms despite compensated hemolysis. |
| Joly et al., JMIR Formative Research 2022 | Web-based cross-sectional patient-reported survey of 50 American patients with self-reported clinician-diagnosed CAD | Before diagnosis, 44/50 (88%) reported symptoms. Common pre-diagnosis symptoms included fatigue/tiredness 37/50 (74%), decreased stamina 23/50 (46%), weakness 22/50 (44%), shortness of breath 19/50 (38%), and acrocyanosis 18/50 (36%). Overall, 45/50 (90%) reported fatigue. During CAD episodes, among 47 respondents, common symptoms included fatigue/tiredness 39/47 (83%), weakness 32/47 (68%), decreased stamina 31/47 (66%), shortness of breath 31/47 (66%), and dark urine 22/47 (47%). Common triggers were cold temperatures 39/47 (83%), winter 32/47 (68%), air conditioning 26/47 (55%), sudden temperature change 20/47 (43%), and infection 15/47 (32%). | Supports patient-reported burden: fatigue, fluctuation, environmental triggers, and lived impact may exceed what hemoglobin alone capture |
These studies should not be read as interchangeable symptom-prevalence estimates. They differ in design, ascertainment, and population. Together, however, they support the same clinical message: CAD presentation is shaped by hemolysis, cold-induced circulatory symptoms, fatigue, environmental fluctuation, and delayed recognition.
Thrombotic complications
Although not a presenting feature in most patients, CAD is associated with thrombotic complications in some cohorts.18 This reinforces a broader framing: CAD is not only a red-cell disease but also has clinically important circulatory and thrombotic associations.19
Practically, when thrombosis occurs in a patient with known or suspected CAD, it should prompt a careful look for concurrent hemolysis and inflammatory or cold-exposure triggers that may have increased disease activity.20
Presentations that delay recognition
CAD is frequently under-recognized because:21
- anemia may be mild or absent early
- symptoms may be attributed to Raynaud phenomenon, aging, or nonspecific fatigue
- laboratory artifacts such as cold-induced red cell clumping may obscure interpretation
Special clinical contexts
Cold exposure during surgery, anesthesia, critical illness, or transfusion may precipitate abrupt hemolysis or symptom worsening, making strict thermal protection clinically essential in high-risk settings.22
Some patients first come to attention when:
- blood typing or crossmatching is complicated
- transfusion responses may be blunted or difficult to interpret during active hemolysis
- blood samples visibly agglutinate at room temperature but normalize after warming; for many clinicians, this is the first clue to CAD
These scenarios reflect temperature-dependent antibody binding and complement-mediated hemolysis characteristic of cold-antibody AIHA.23
Clinical History: Questions That Clarify Pattern
Ask about:
- duration and tempo of symptoms (gradual vs abrupt)
- seasonal pattern or cold-triggered fluctuation
- color change pattern (persistent cyanosis vs triphasic Raynaud)
- dark urine episodes, especially after cold exposure or infection
- transfusion history, including unexpected response
- recent infection (especially 1–3 weeks prior in younger patients)
- constitutional symptoms suggesting lymphoproliferative disease
- prior “Raynaud” label with fatigue out of proportion to hemoglobin
Physical Examination
Examine for:
General
• pallor
• mild jaundice or scleral icterus
Acral Findings
• persistent dusky acrocyanosis
• mottled cold-induced discoloration that improves with warming
• absence of classic triphasic Raynaud pattern
Severe Hemolysis (less common)
• tachycardia
• signs of heart failure
• hepatosplenomegaly (splenomegaly is not a dominant feature in most patients with primary CAD)
Laboratory Artifacts
Check for:
• visible red cell agglutination at room temperature
• spuriously elevated MCV or MCHC
• RBC count falsely low
• correction of indices after warming sample
Helpful Clinical Terms
Block content:
Acrocyanosis
Persistent bluish or dusky discoloration of acral areas such as fingers, toes, ears, or nose. In CAD, it reflects cold-induced red-cell agglutination and impaired microvascular flow more than classic vasospasm.
Raynaud phenomenon
Episodic vasospasm of small arteries, classically producing triphasic color change: white, then blue, then red. CAD can mimic Raynaud phenomenon, but discoloration may be more persistently dusky or cyanotic and improve primarily with warming.
Livedo-like discoloration
A mottled, netlike violaceous pattern of skin discoloration caused by uneven blood flow through the cutaneous microcirculation. In CAD, livedo-like changes may appear or worsen with cold exposure and improve with warming.
Acral
Refers to peripheral body parts, especially fingers, toes, ears, and nose, where cooling is most pronounced and cold-induced symptoms are often most visible.
These terms describe patterns of appearance and physiology. In CAD, the key question is whether cold-induced red-cell agglutination is contributing to impaired flow.
Explicit principle
CAD rarely announces itself with a single dramatic finding. Recognition depends on noticing parallel physiology, temperature dependence, and discordance between laboratory values and lived experience.
Key Points
- parallel streams: CAD produces hemolytic and circulatory symptom streams shaped by temperature and blood flow.
- symptoms may exceed anemia: circulatory symptoms and fatigue may be more disabling than anemia itself.
- environment matters: symptoms fluctuate with environmental temperature and physiologic stressors.
- hemoglobin is incomplete: hemoglobin level alone is an unreliable measure of disease severity.
Reflect and Apply
A 67-year-old woman presents in late January with fatigue, mild anemia (hemoglobin 10.1 g/dL), and three years of cold-sensitive fingers she has attributed to “Raynaud’s.” She reports that her fingers turn dusky blue in grocery store freezer aisles and improve with warming. She denies dramatic color cycling. Her fatigue feels out of proportion to her laboratory values.
Before ordering additional tests, pause.
- Which symptom stream appears dominant: hemolysis-driven, agglutination-driven, or both?
- What bedside features help distinguish CAD-related circulatory symptoms from primary Raynaud phenomenon?
- Does the hemoglobin level alone capture her disease burden?
- What environmental triggers would you specifically ask about?
Now consider a different patient with hemoglobin 8.8 g/dL, no circulatory symptoms, and minimal fatigue discovered incidentally during routine labs.
- Which symptom stream is dominant?
- How might the clinical picture differ despite a lower hemoglobin?
- What additional history would help clarify disease activity?
Test your thinking
A short, judgment-focused quiz on clinical presentation of cold agglutinin disease.
