Why cold agglutinin–mediated hemolysis is best understood as a spectrum
Note: The video and audio linked above were generated with the assistance of AI. Clinical accuracy has been reviewed, but no AI-generated content can be guaranteed to be fully error-free.
Why this spoke matters
Cold agglutinin disease is often taught as a categorical diagnosis: present or absent, primary or secondary. The definitions matter, but real patients may test the edges of those definitions. Categories are useful, but the underlying biology is graded, dynamic, and context dependent.
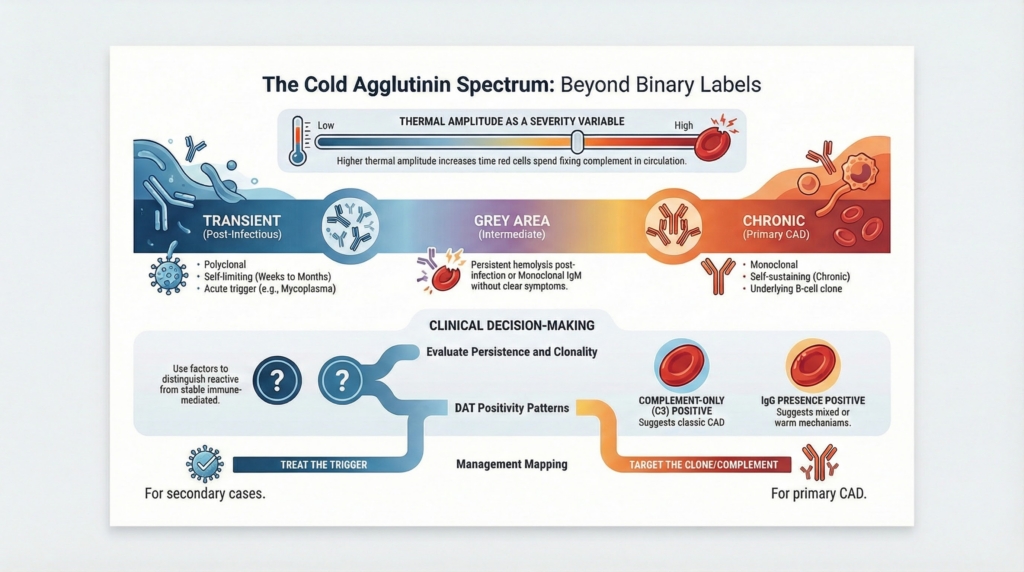
Cold agglutinin–mediated disorders exist across a biologic spectrum, from incidental or transient cold agglutinins to secondary cold agglutinin syndromes and primary CAD; primary CAD remains a distinct clinicopathologic entity within that spectrum. Across this spectrum, clinical expression is shaped by antibody characteristics, complement activity, clonal persistence, thermal amplitude, and environmental exposure.1
The direct antiglobulin test (DAT) is the most practical laboratory correlate of clinically significant cold antibody–mediated hemolysis. In classic cold antibody–mediated hemolysis, the DAT is typically complement-positive, with absent or minimal IgG. Strong DAT IgG positivity shifts the differential toward warm AIHA or a mixed AIHA phenotype and should change how the hemolytic mechanism is interpreted.2
In classic primary cold agglutinin disease (CAD), hemolysis is primarily complement-mediated. Across the broader spectrum, mixed or overlapping mechanisms may be present.
Classification is not an academic exercise. It explains prognosis, predicts treatment response, and clarifies why relapse patterns differ so widely between patients who may look similar at presentation.3
This spoke moves beyond binary thinking by introducing a biologically graded framework for distinguishing incidental or transient cold agglutinins, secondary cold agglutinin syndrome, primary CAD, and mixed phenotypes, while preserving the clinicopathologic identity of primary CAD.
Cold agglutinin-mediated disease exists on a continuum
Cold agglutinins themselves are common. What varies is not their presence, but their behavior, persistence, and biological consequences.4
Across patients, there is continuous variation in:5
- antibody clonality
- thermal amplitude
- efficiency and durability of complement activation
- persistence over time
- relationship to an underlying B-cell clone
At one end of the spectrum are transient, polyclonal cold agglutinins that appear during infection and disappear without consequence.6
At the other end is primary cold agglutinin disease, driven by a stable clonal B-cell disorder with chronic, self-sustaining complement-mediated hemolysis.7
Between these poles lies an intermediate territory that may not fit cleanly into a single label at initial presentation.8
Examples of this intermediate territory that clinicians actually encounter include:9
- persistent hemolysis months after an apparent post-infectious episode
- monoclonal IgM with minimal symptoms and unclear clinical significance
- a classic CAD phenotype in which a clone is not demonstrable on initial evaluation
These patterns reflect biology unfolding over time rather than fixed categories at presentation.10
Clinical orientation points
Broad patterns across populations include:
- cold agglutinins may be detectable at low titers in many healthy individuals
- infection-associated cold agglutinins are often polyclonal and usually self-limited
- monoclonal cold agglutinins are present in most patients with primary CAD
- many patients with primary CAD lack overt clinical or radiologic lymphoma at diagnosis
- higher thermal amplitude generally correlates with greater clinical significance
These patterns orient interpretation; they do not define diagnostic thresholds.
Why classification matters clinically
Classification should not be treated as a label. It is a tool for alignment: matching expected biology to proportionate evaluation and therapy.11
Classification helps predict:12
- whether hemolysis will persist or resolve
- whether cold avoidance alone is sufficient
- whether relapse is likely after treatment
- whether clone-directed therapy is required for durable control
Misclassification cuts both ways:13
- labeling a persistent clonal process as “benign cold agglutinins” can lead to years of untreated morbidity
- labeling a self-limited post-infectious process as “primary CAD” can drive unnecessary invasive evaluation or therapy
Primary (clonal) cold agglutinin disease
Primary CAD represents the stable end of the spectrum.14
It is characterized by:15
- monoclonal IgM cold agglutinins
- antibody features that confer clinical significance, such as higher thermal amplitude
- chronic, complement-mediated hemolysis
- an underlying clonal B-cell lymphoproliferative disorder, usually confined to the bone marrow
A clonal B-cell population is demonstrable in most cases, but failure to detect one initially does not exclude the diagnosis.
The associated clonal disorder is typically low-grade and indolent. Clone size often correlates poorly with anemia severity.16
This apparent disconnect reflects the fact that antibody quality matters more than antibody quantity. Thermal amplitude is one way to name that quality: the higher the thermal amplitude, the more time red cells spend with IgM binding (and complement being fixed) in physiologic circulation, which increases the likelihood of clinically important complement deposition and sustained extravascular hemolysis.1718
Primary CAD behaves as a chronic clonal, complement-mediated hemolytic disorder, not a transient reactive phenomenon.19
This biology explains why:20
- corticosteroids are usually ineffective
- elapse is common when the underlying clonal or complement biology is not addressed
- durable control usually requires clone-directed and/or complement-directed therapy
Secondary cold agglutinin syndromes
Secondary cold agglutinin syndromes occupy a different region of the spectrum. They are defined not only by antibody presence, but by antibody source, thermal behavior, clinical context, and trajectory.21
Typical features include:22
- polyclonal or transient cold agglutinins
- often lower thermal amplitude
- hemolysis temporally linked to another condition
- improvement with treatment or resolution of the underlying trigger
Common associations include:23
- acute infections, especially Mycoplasma pneumoniae and Epstein–Barr virus
- autoimmune disease
- overt lymphoid malignancy
A practical refinement is important: “secondary to infection” and “secondary to lymphoma” are not the same clinical problem. Infection-associated disease is often self-limited, while malignancy-associated cold agglutinin hemolysis may persist or relapse in parallel with the neoplasm.24
Persistence should prompt reevaluation. Persistent or recurrent hemolysis in an apparent secondary cold agglutinin syndrome does not automatically mean primary CAD, but it changes the question: is this a prolonged reactive process, ongoing autoimmune or inflammatory disease, malignancy-associated cold agglutinin hemolysis, or an emerging clonal cold agglutinin disorder? Classification may need to be revisited as the clinical trajectory becomes clearer over time.25
Post-infectious cold agglutinins: where they fit, and where they do not
Post-infectious cold agglutinins deserve special attention because they are a frequent source of diagnostic confusion. They are common, expected, and usually benign.26
Typical features of post-infectious cold agglutinins include:27
- appearance during or shortly after infection
- polyclonal antibodies
- usually low thermal amplitude
- resolution over weeks to months
- rare need for disease-specific therapy
What they are not:28
- primary CAD
- a chronic lymphoproliferative disorder
- a reliable predictor of long-term hemolysis in most patients
Post-infectious cold agglutinins are common and often benign, but that pattern should not be generalized to all cold agglutinins. Persistence beyond the expected infectious period, especially with ongoing hemolysis or features suggesting higher thermal amplitude, should prompt reassessment for a persistent reactive process or an emerging clonal cold agglutinin disorder.29
Mixed phenotypes: a boundary worth naming
Some patients do not fit cleanly into ‘warm’ or ‘cold’ categories, such as those with DAT positivity for both IgG and C3 and features suggesting dual mechanisms. These mixed phenotypes require special caution because the treatment logic is not identical to classic primary CAD: clinicians often need to evaluate and treat both components, rather than assuming that a single downstream target (for example, complement alone) will capture the entire biology.30
They represent boundary conditions rather than central exemplars of cold agglutinin disease, and they should not be forced into a classic CAD framework simply because C3 is present.
Spectrum thinking in practice
Rather than asking “Is this CAD, yes or no?”, ask questions that locate the patient along the spectrum. Start with the primary discriminators, persistence, clonality, and clinical context, then use modifiers such as thermal amplitude and complement activity to refine risk and expectations. Evidence of a clonal B-cell population strongly supports primary CAD, but absence of a detectable clone at presentation does not fully exclude it.31
The following questions help locate the patient along that spectrum:
- Is hemolysis transient or persistent?
Persistence distinguishes self-limited cold agglutinin syndromes from chronic clonal disease and often only becomes clear with time and follow-up. - Is the antibody polyclonal or monoclonal?
Polyclonal antibodies are typical of post-infectious disease, whereas monoclonality supports primary CAD, even when marrow findings are subtle. - Does disease activity track with infection, season, or neither?
Temporal association with infection or resolution with recovery favors secondary disease, while season-independent or progressive hemolysis raises concern for clonal persistence. - Is complement activation episodic or sustained?
Chronic complement-mediated hemolysis is a defining biological feature of primary CAD, whereas episodic activation is more typical of secondary or post-infectious states. - Is there evidence of an underlying clonal B-cell population, now or over time?
Failure to detect a clone at presentation does not exclude primary CAD, and reassessment may be required as the disease trajectory becomes clearer over time.
Thermal amplitude is best treated as a severity-shaping variable across the spectrum, not a membership card for a single category. It can intensify symptoms at any position, while clonality and persistence determine whether the disease endures.32
Treatment implications, mapped to spectrum position
This is not a detailed treatment section. It is a classification-to-expectations map.33
- Post-infectious cold agglutinins:
Disease is usually self-limited. Supportive care and time are often sufficient, and disease-specific therapy is rarely required. - Secondary to a trigger (infection, autoimmune disease):
Management is primarily directed at the underlying condition, with reassessment over time to ensure that hemolysis resolves rather than persists. - Secondary to lymphoma:
Hemolysis often tracks with the course of the malignancy, and durable control typically depends on effective treatment of the underlying lymphoid disorder. - Primary clonal cold agglutinin disease:
The biology is chronic and self-sustaining. Meaningful and durable control usually requires clone-directed and/or complement-directed therapy rather than nonspecific immunosuppression.
Explicit principle
Avoid forcing continuous biology into oversimplified boxes; use labels only when they clarify mechanism, prognosis, and treatment.
Cold agglutinin–mediated disease is not defined by a single threshold or test result. It evolves over time, responds to context, and reflects an interaction between immune biology and environment. Classification should illuminate that complexity, not obscure it.
Test your thinking
A short, judgment-focused quiz on classification of cold agglutinin disease.
Key terms used in this essay
Primary CAD:
chronic clonal cold-antibody hemolytic anemia driven by monoclonal IgM and complement activation.
Secondary cold agglutinin syndrome:
cold agglutinin-mediated hemolysis occurring in association with infection, autoimmune disease, or overt lymphoid malignancy.
Transient cold agglutinins:
usually low-titer, often polyclonal antibodies that arise temporarily, often after infection, and may cause little or no hemolysis.
