When Two Treatment Strategies Meet
When Complement Inhibition Is Not Enough
Choosing between complement blockade and clone-directed therapy in cold agglutinin disease
By William C. Aird, MD
Case Presentation
A 60-year-old man with cold agglutinin disease (CAD) presents to hematology clinic for routine follow-up.
Three years earlier he had developed fatigue and jaundice and was found to have hemolytic anemia.
Initial laboratory evaluation demonstrated:
| Test | Result |
|---|---|
| Hemoglobin | 6.7 g/dL |
| Total bilirubin | 3.5 mg/dL |
| LDH | 312 U/L |
| Haptoglobin | undetectable |
Direct antiglobulin testing showed:
- DAT positive
- anti-complement positive
- anti-IgG negative
Cold agglutinin titer was 1:10,240.
Peripheral smear demonstrated red cell agglutination.
These findings established the diagnosis of cold agglutinin disease.
Underlying Biology
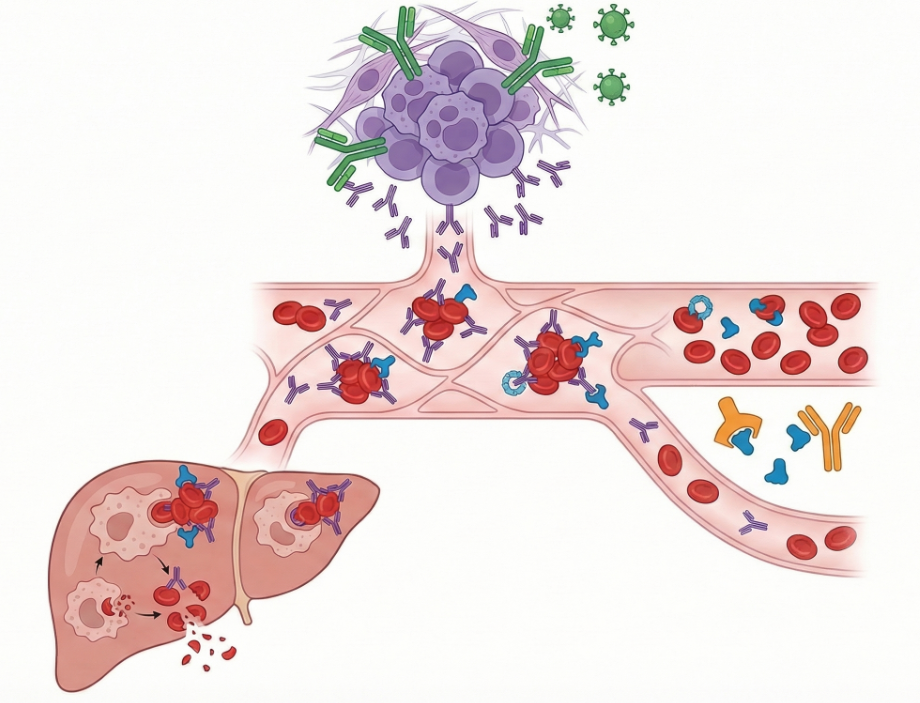
Bone marrow biopsy revealed a small B-cell lymphoproliferative clone.
Flow cytometry showed kappa-restricted B cells lacking CD5 and CD10, consistent with a primary CAD-associated lymphoproliferative disorder.
Such indolent B-cell clones share features with lymphoplasmacytic or marginal zone lymphomas and are responsible for production of the pathogenic monoclonal IgM.
These antibodies bind red cells and activate complement.
Which mechanism is primarily responsible for hemolysis in cold agglutinin disease?
Explanation
Cold agglutinin disease is caused by monoclonal IgM antibodies that bind red cells at lower temperatures.
These antibodies activate the classical complement pathway, leading to:
- C3 deposition on red cells
- extravascular hemolysis in the liver
- occasional complement-mediated intravascular hemolysis
Because the process is complement-mediated, therapies targeting complement can rapidly improve hemolysis.
Initial Treatment
During the winter of 2023 the patient developed worsening cold-induced symptoms and fatigue.
Hemoglobin fell into the 6–7 g/dL range, and treatment was initiated with sutimlimab, a monoclonal antibody that inhibits C1s, blocking activation of the classical complement pathway.
Cold agglutinin antibodies bind red cells more avidly at lower temperatures, so hemolysis and symptoms often worsen during colder months and improve during warmer seasons.
After several infusions, his hemoglobin improved dramatically:
| Date | Hemoglobin |
|---|---|
| Before treatment | 6.7 g/dL |
| After 2 months | 10.6 g/dL |
| After 3 months | 12.8 g/dL |
Hemolysis markers normalized and symptoms resolved.
A patient with CAD experiences rapid improvement in hemoglobin after starting sutimlimab.
Which of the following laboratory findings would most likely remain abnormal despite treatment?
Explanation
Sutimlimab blocks C1s, a key enzyme in the classical complement pathway.
This prevents complement activation on red cells and rapidly halts complement-mediated hemolysis.
However, complement inhibition does not eliminate the underlying B-cell clone that produces the pathogenic IgM antibody.
As a result, cold agglutinin titers typically remain detectable, even when hemolysis and anemia improve.
Clinical Course
The patient received six doses of sutimlimab with excellent hematologic response.
Treatment was then stopped during the summer to determine whether hemolysis would remain controlled without therapy.
Shortly afterward he developed pneumonia and viral infection, and hemoglobin fell again into the 8 g/dL range, indicating recurrent hemolysis.
Infection itself can amplify complement activation in patients with CAD. The relapse therefore likely reflected a combination of stopping complement inhibition and infection-driven complement activation.
Because hemolysis recurred, clinicians elected to treat the underlying B-cell clone.
What is the rationale for treating the B-cell clone in primary cold agglutinin disease?
Explanation
In primary cold agglutinin disease, the pathogenic antibodies are produced by a clonal B-cell population.
Clone-directed therapy aims to:
- reduce or suppress the B-cell clone
- decrease production of IgM cold agglutinins
- achieve durable remission of hemolysis
This approach addresses the root cause of the disease, whereas complement inhibition controls downstream effects.
Clone-Directed Therapy
The patient began bendamustine plus rituximab (BR) therapy.
| Cycle | Date |
|---|---|
| Cycle 1 | Aug 2023 |
| Cycle 2 | Sep 2023 |
| Cycle 3 | Oct 2023 |
| Cycle 4 | Nov 2023 |
Over the following months hemoglobin steadily improved.
Hemoglobin rose from approximately 8 g/dL before treatment to the 11–12 g/dL range, accompanied by normalization of hemolysis markers.
By early 2024 he achieved a durable hematologic response.
Current Status
At his most recent visit he reports feeling well and has adopted behavioral strategies to minimize cold exposure.
He has stopped coaching youth hockey because of prolonged cold exposure but occasionally attends games.
Current laboratory values:
| Test | Result |
|---|---|
| Hemoglobin | 12.4 g/dL |
| LDH | normal |
| Bilirubin | normal |
| Haptoglobin | detectable |
These findings suggest near-complete remission of hemolysis.
He is currently managed with active surveillance.
If hemolysis recurs in the future, treatment options would include repeat clone-directed therapy or re-initiation of complement inhibition.
Which statement best summarizes the relationship between complement inhibition and clone-directed therapy in CAD?
Explanation
Complement inhibitors such as sutimlimab act downstream, preventing complement-mediated destruction of red cells.
Clone-directed therapies such as rituximab-based regimens act upstream, reducing production of pathogenic IgM antibodies.
Thus:
- complement inhibition → rapid control of hemolysis
- clone-directed therapy → potential for long-term remission
Many patients receive both approaches sequentially during the course of their disease.
Teaching Points
- Cold agglutinin disease is driven by monoclonal IgM antibodies produced by a clonal B-cell population.
These antibodies activate complement and lead to complement-mediated hemolysis. - Complement inhibition rapidly controls hemolysis in CAD.
Agents such as sutimlimab block the classical complement pathway but do not eliminate the underlying clone. - Clone-directed therapy targets the underlying B-cell lymphoproliferative disorder.
Regimens such as bendamustine plus rituximab can produce durable remissions. - Patients may transition between therapeutic strategies over the course of disease.
Complement inhibition provides rapid control, while clone-directed therapy offers the possibility of long-term disease modification.
Why This Case Matters
Cold agglutinin disease illustrates two fundamentally different therapeutic strategies.
One approach targets the mechanism of hemolysis by blocking complement activation.
The other targets the source of the pathogenic antibody by suppressing or eliminating the B-cell clone.
Complement inhibition can produce rapid control of hemolysis, often within weeks.
Clone-directed therapy acts more slowly but may provide longer-lasting remission.
Clinical management often involves sequencing these strategies over time, using complement inhibition for rapid stabilization and clone-directed therapy for durable disease control.
