When “Macrocytic Anemia” Was Something Else
When “Macrocytic Anemia” Was Something Else
Cold agglutinin disease evolving over time
By William C. Aird, MD
Case Presentation
A 70-year-old man with a history of:
- prostate cancer
- coronary artery disease with prior STEMI
- seizure disorder
- peripheral neuropathy
was referred for evaluation of new macrocytic anemia.
He reported several new symptoms over the prior months:
- feeling persistently cold
- blue discoloration of the fingers in cold weather
- constipation
- weight loss during cardiac rehabilitation
Laboratory testing showed:
| Test | Result |
|---|---|
| Hemoglobin | 11.5 g/dL (baseline 13.5) |
| MCV | 101 fL |
| Reticulocytes | 7.5% (absolute 0.22 ×10⁶/µL) |
| Haptoglobin | undetectable |
| Ferritin | 213 ng/mL |
| B12 | normal |
| Folate | normal |
Although the MCV was mildly elevated, macrocytosis in this setting was most consistent with reticulocytosis from hemolysis rather than nutritional deficiency, given the normal B12 and folate levels.
Inflammatory markers were markedly elevated:
- ESR: 103
- CRP: 13.4
Which of the following symptoms is most suggestive of cold agglutinin disease?
Explanation
Cold agglutinin disease often produces circulatory symptoms caused by agglutination of red cells in cooler peripheral tissues.
Common symptoms include:
- acrocyanosis
- Raynaud-like changes
- livedo reticularis
These symptoms may precede the diagnosis of hemolytic anemia.
Diagnostic Workup
Further evaluation demonstrated:
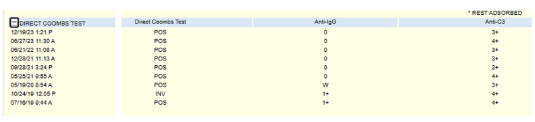
- direct antiglobulin test positive for C3
- additional evidence of hemolysis
- red cell agglutination on smear
DAT results:
A bone marrow biopsy showed:
- mildly hypercellular marrow
- erythroid predominance
- small lambda-restricted B-cell population

What is the most common underlying hematologic process in primary cold agglutinin disease?
Explanation
Most patients with primary CAD harbor a small clonal B-cell population in the bone marrow.
These clones produce monoclonal IgM antibodies that:
- cause red cell agglutination in cooler peripheral tissues
- activate complement on the red cell surface
- drive chronic hemolysis
However, the clone often does not meet diagnostic criteria for overt lymphoma.
Clinical Course
Several months after diagnosis, the patient experienced a severe hemolytic episode after cold exposure.
He presented with:
- hematocrit of 17%
- marked red cell agglutination
- need for multiple blood transfusions
Treatment with rituximab was initiated.
He received four weekly doses, resulting in:
- improvement in hemoglobin
- reduction in hemolysis markers
- improvement in fatigue and exercise tolerance
Cold agglutinin disease can be treated by targeting either the pathogenic antibody–producing B-cell clone or the complement cascade that mediates hemolysis. What is the primary mechanism by which rituximab improves cold agglutinin disease?
Explanation
Rituximab targets CD20 on B cells, reducing production of the pathogenic IgM cold agglutinin.
This decreases complement activation and hemolysis.
Clinical improvement often occurs several weeks after treatment, as circulating IgM antibodies gradually decline following B-cell depletion.
A Later Development
Several years later, the patient was diagnosed with systemic lupus erythematosus (SLE) based on:
- positive ANA
- anti-SSA antibodies
- anti-Smith antibodies
- anti-RNP antibodies
In retrospect, the markedly elevated ESR and CRP at the time of initial presentation may have reflected early systemic immune activation, preceding the eventual diagnosis of autoimmune disease.
With treatment of SLE using mycophenolate, his hemoglobin stabilized and markers of hemolysis resolved.
His cold agglutinin disease was therefore reclassified as secondary to autoimmune disease.
This evolution illustrates an important diagnostic reality:
patients who initially appear to have primary CAD with a small B-cell clone may later prove to have a systemic autoimmune driver of hemolysis.
Teaching Points
- Cold agglutinin disease may initially appear as macrocytic anemia due to reticulocytosis.
Reticulocytes are larger than mature red cells, so increased reticulocyte production during hemolysis can elevate the measured MCV. - Circulatory symptoms such as acrocyanosis or Raynaud-like changes can provide important early clues.
Red cell agglutination occurs in cooler peripheral tissues, producing characteristic cold-induced discoloration of the extremities. - Many patients with CAD have an underlying clonal B-cell population in the bone marrow.
These clones produce monoclonal IgM antibodies that bind red cells and trigger complement-mediated hemolysis. - Rituximab therapy improves CAD by targeting B cells producing pathogenic IgM antibodies.
B-cell depletion reduces antibody production, leading to decreased complement activation and gradual improvement in hemolysis.
Why This Case Matters
Cold agglutinin disease is often a chronic, evolving disorder.
Patients may move through several stages:
- initial unexplained anemia
- recognition of hemolysis
- identification of a B-cell clone
- treatment with B-cell–directed therapy
- discovery of an underlying systemic disease
This case illustrates that diagnostic labels may evolve over time, and that conditions initially interpreted as primary CAD may ultimately prove secondary to systemic autoimmune disease.
Recognizing this trajectory helps clinicians revisit earlier assumptions and refine the diagnosis as new information emerges.
