D-Dimers
Prev
1 / 4 Next
Prev
1 / 4 Next
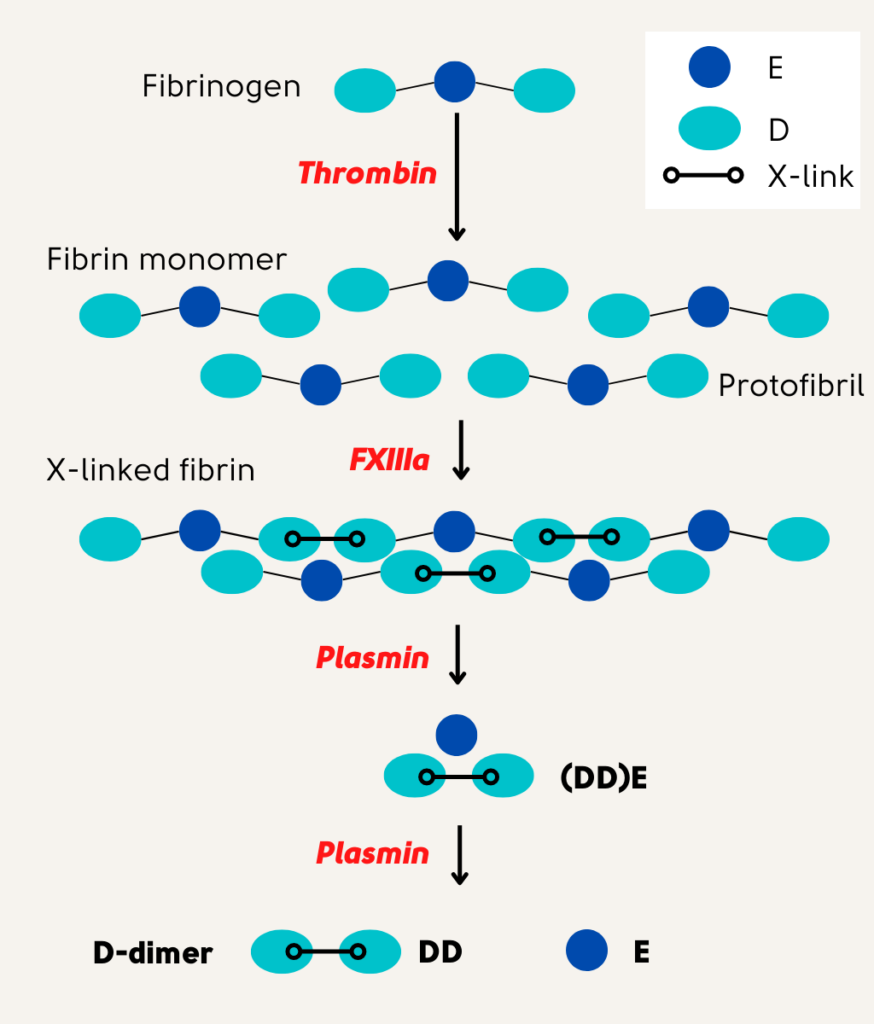
D-dimer assay:
- D-dimer is detected and quantified in whole blood, plasma, or serum using monoclonal antibodies that recognize a specific epitope on cross-linked D-dimer molecules that are otherwise absent on the D-domain of fibrinogen and fibrin monomers that are non-cross-linked.
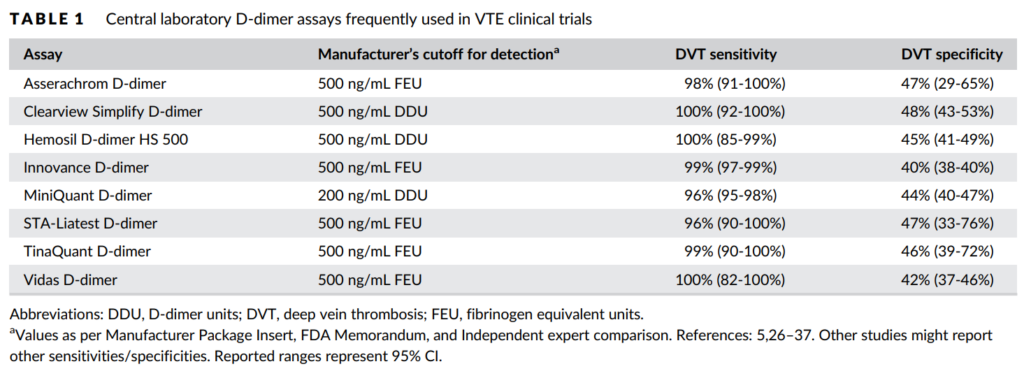
- > 30 commercial D-dimer assays are available, including
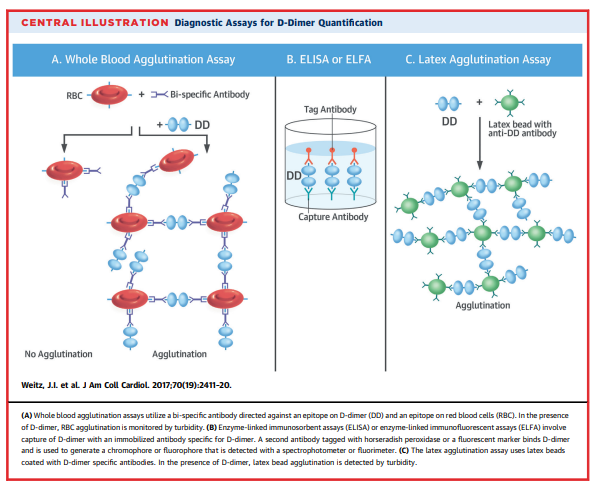
- Enzyme-linked immunosorbent assays (ELISA)
- Immunofluorescent assays
- Latex agglutination assays
Clinical uses of D-dimer:
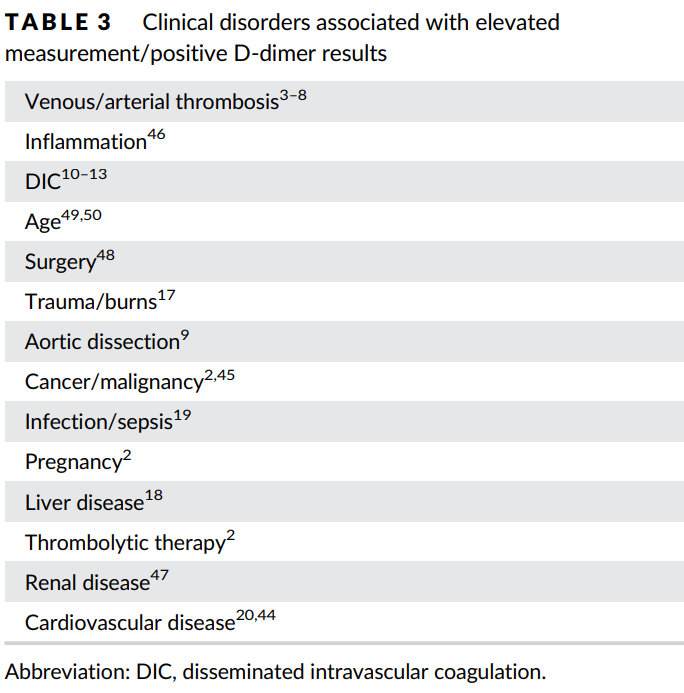
- Exclusion of venous thromboembolism
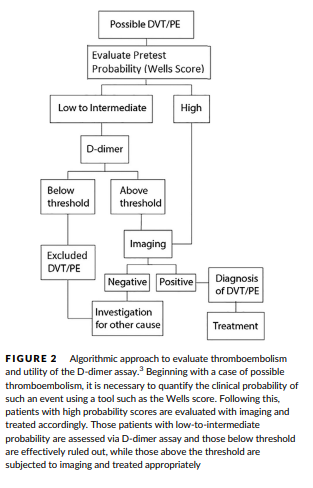
- Assessment of the duration of anticoagulation
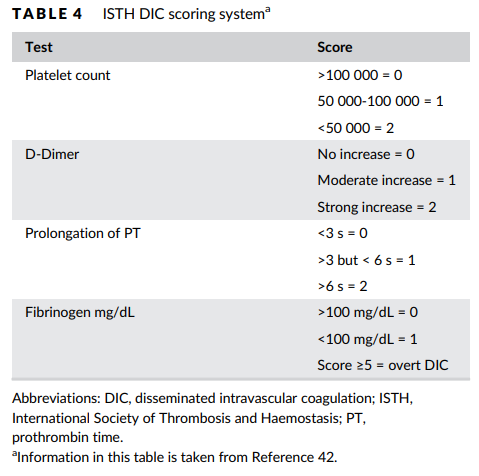
- Diagnosis of disseminated intravascular coagulation
Prev
1 / 4 Next
