Study Design
Prev
1 / 0 Next
Prev
1 / 0 Next
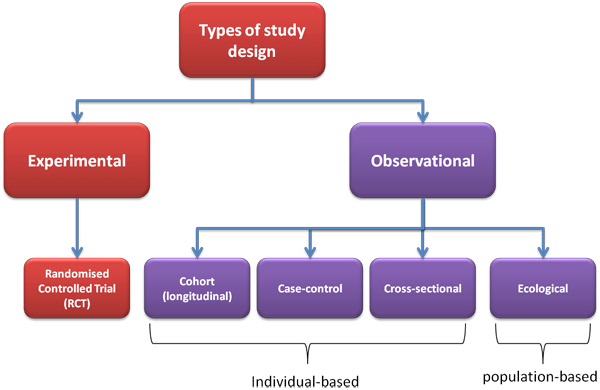
- Multicenter, randomized, controlled, open-label clinical trial
- n = 195
- High-dose dexamethasone: 95
- Prednisone: 97
- Setting: Nine study sites in China
- Enrollment period: January 2011 and May 2014
- Follow-up period: 6 months
- Analysis: Intention-to-treat
- Primary efficacy outcomes: Overall response (OR) and sustained response (SR) rates
- OR = complete response (CR) + response
- CR: Platelet count > 100,000 and absence of bleeding
- Response: Platelet count > 30,000 and at least a two-fold increase from baseline and absence of bleeding
- SR: Platelet count maintained > 30,000 with absence of bleeding symptoms and no requirement for additional ITP treatment for 6 months following achievement of initial response
- Primary safety outcome: Adverse events

Prev
1 / 0 Next
